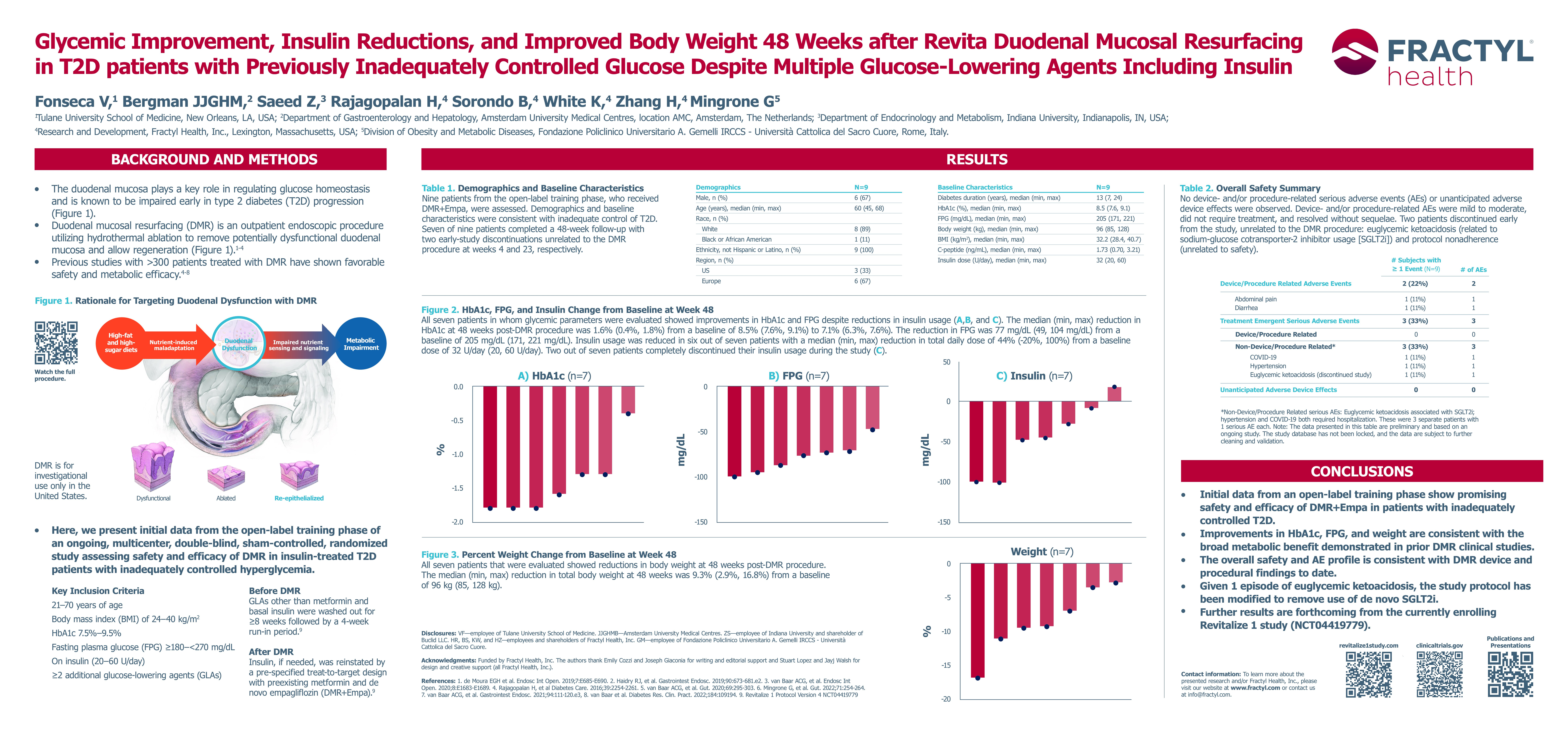
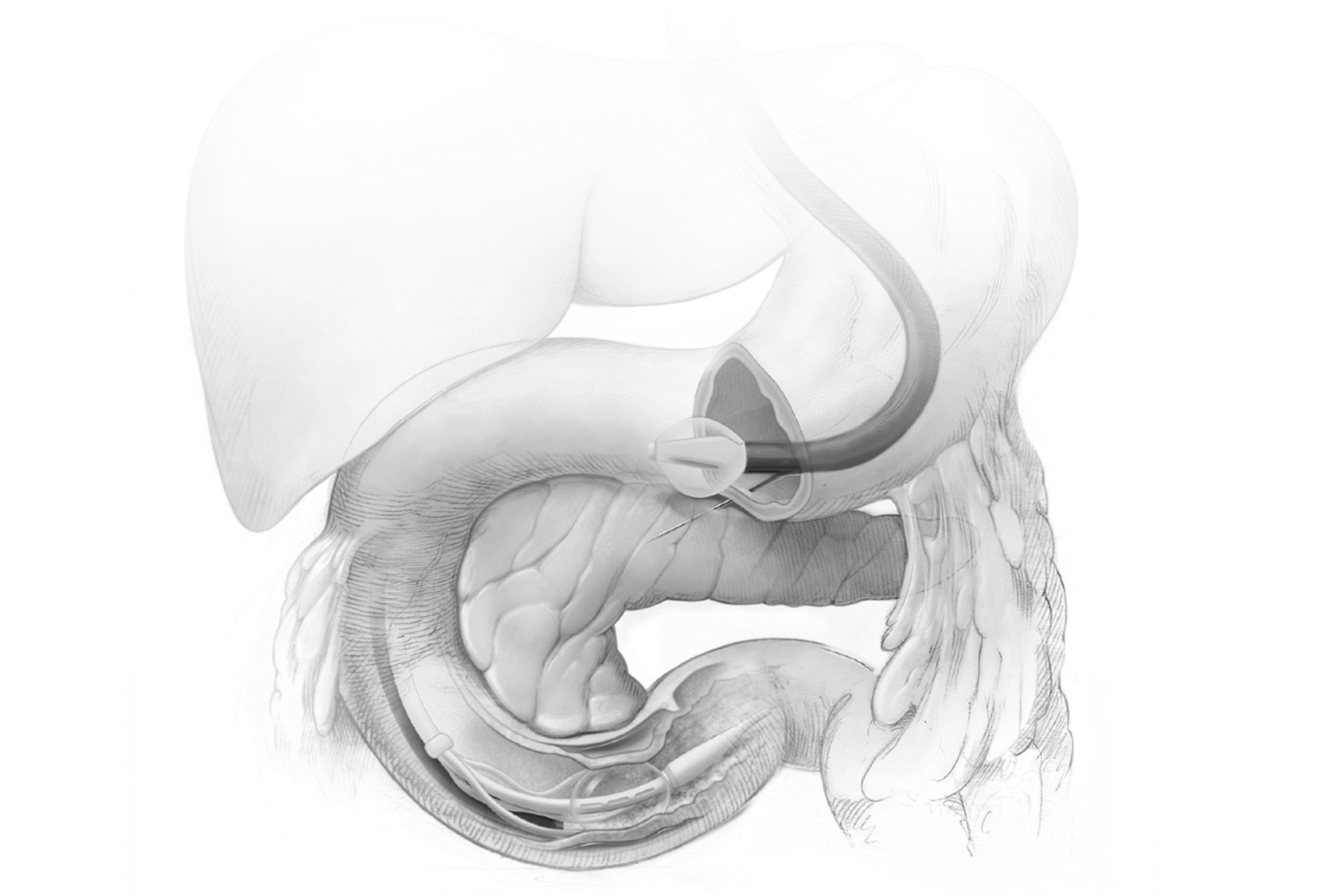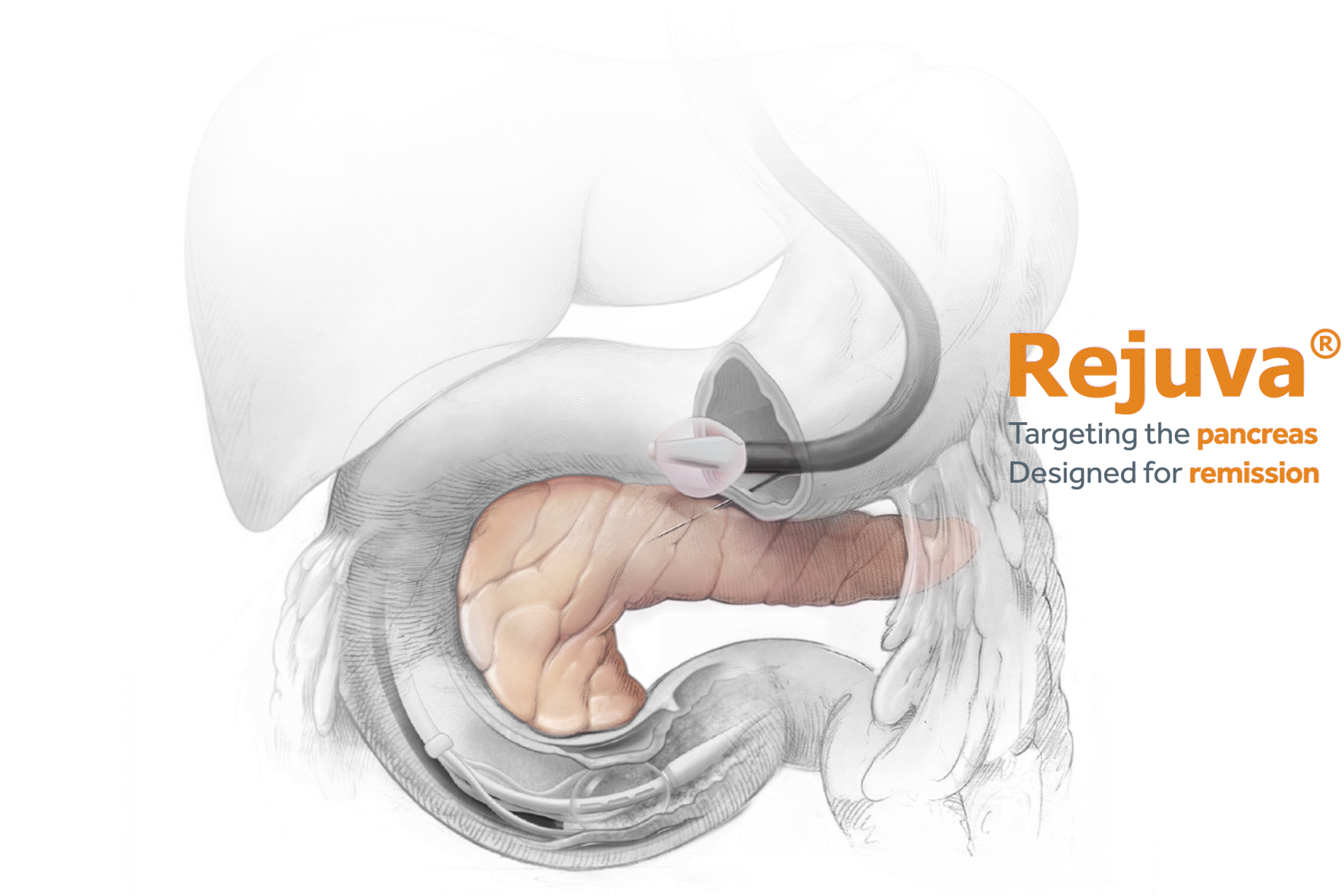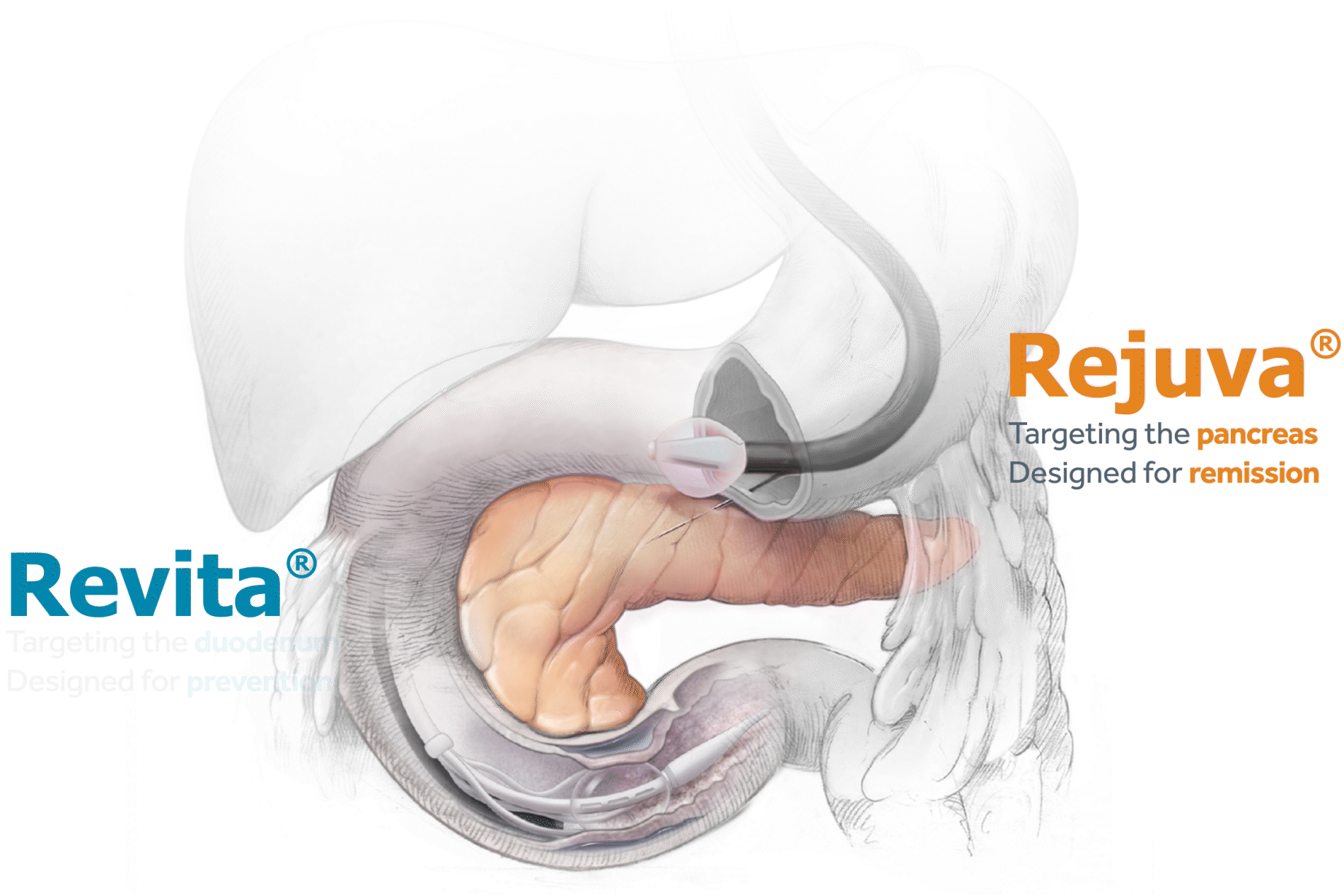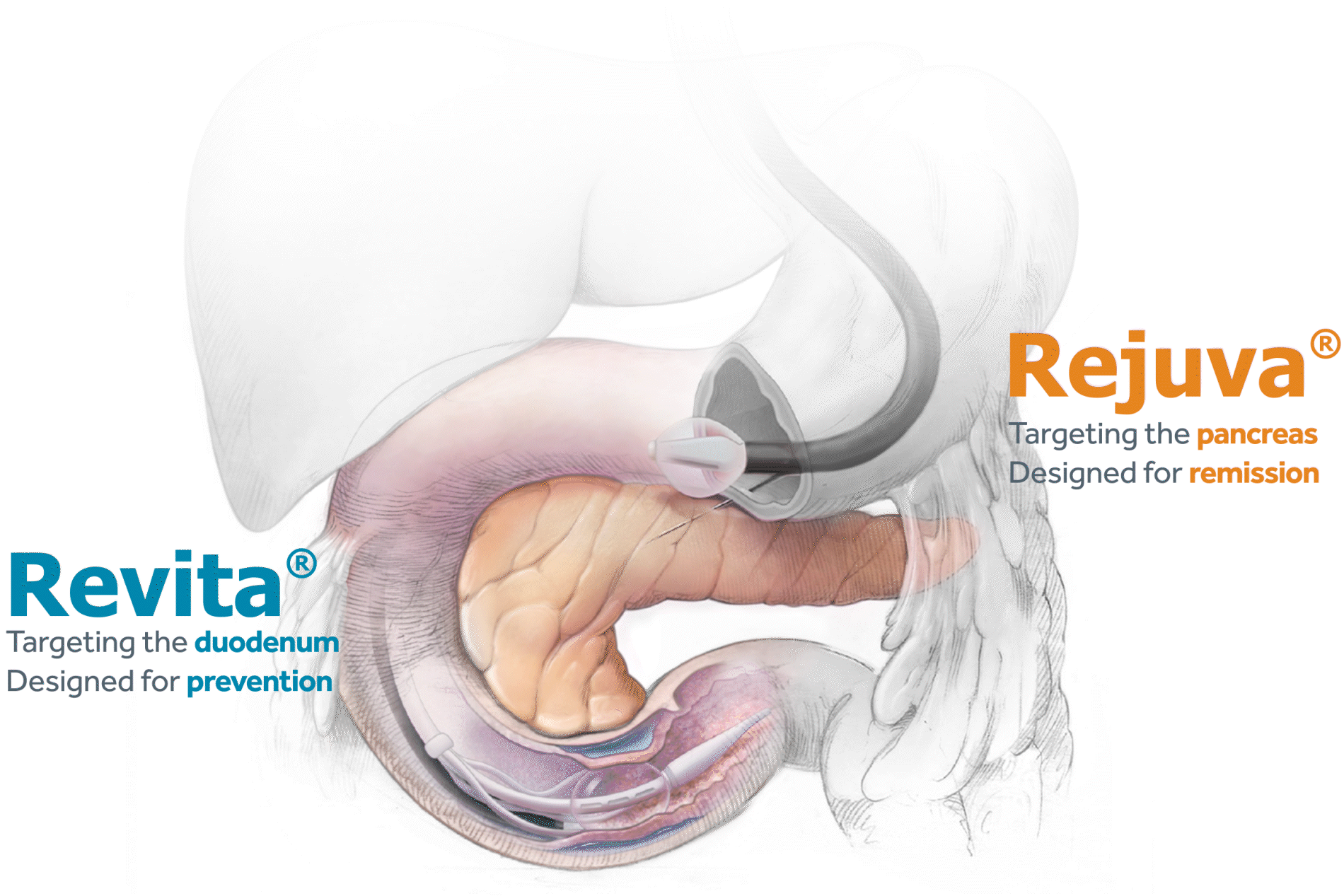
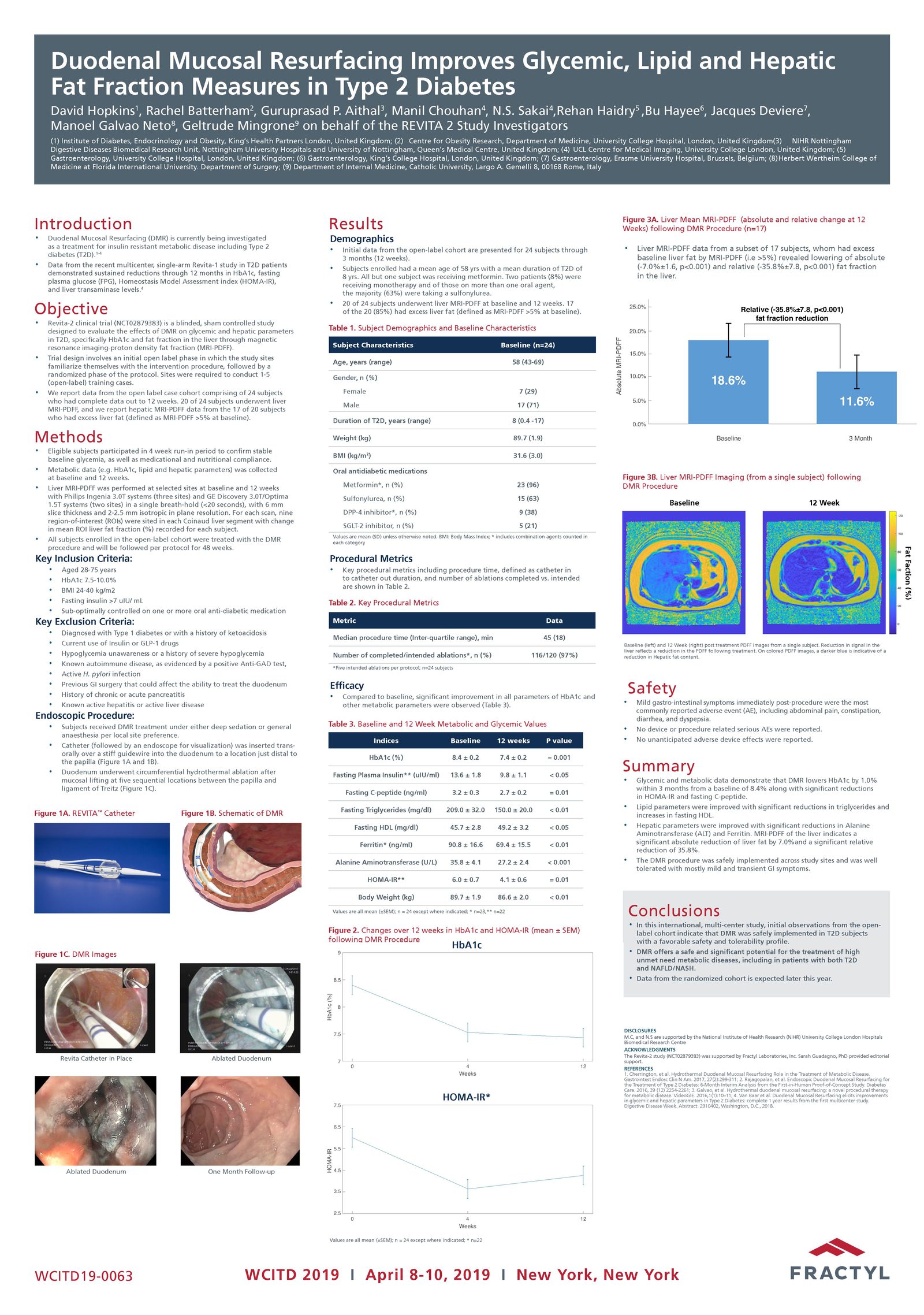
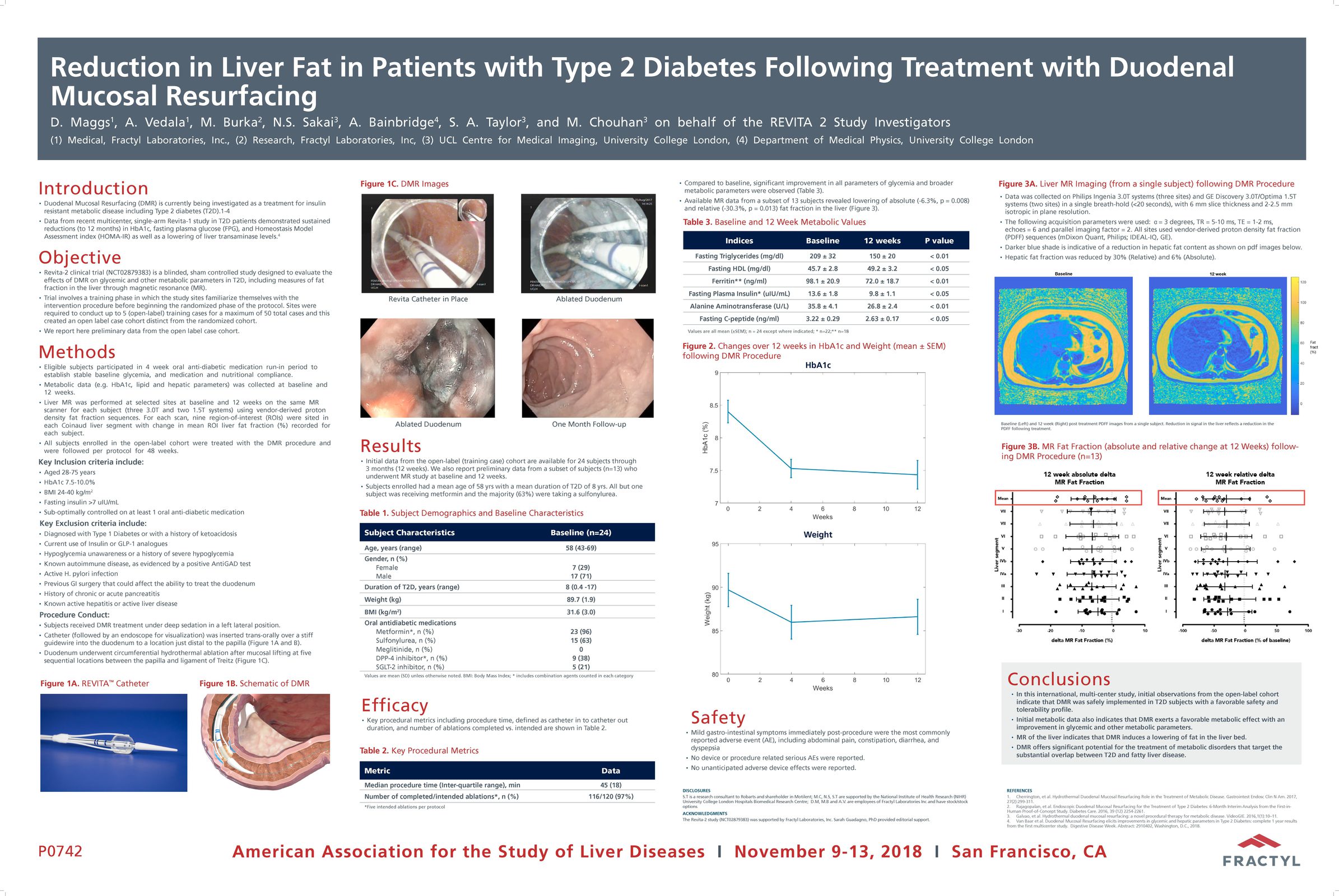
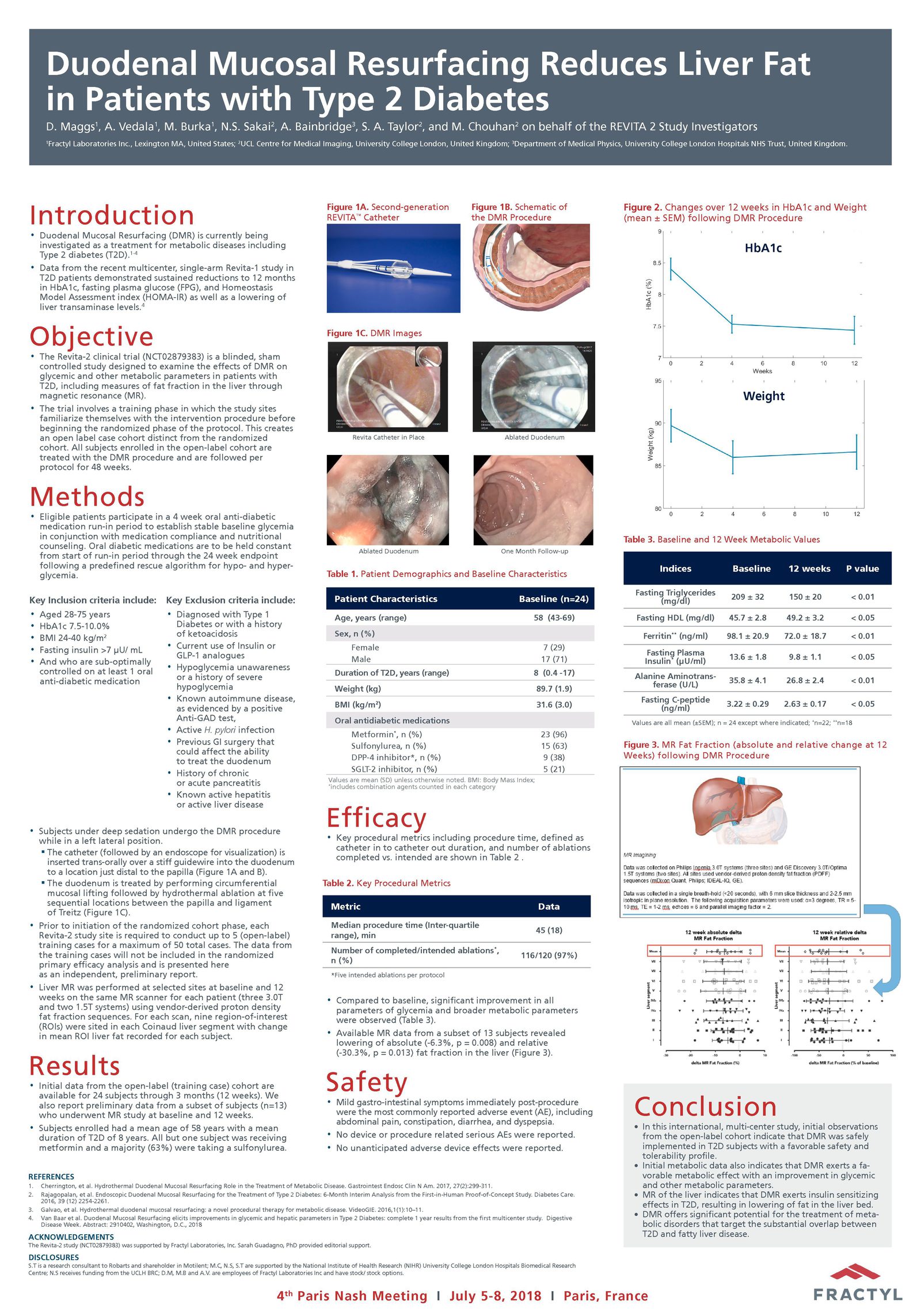
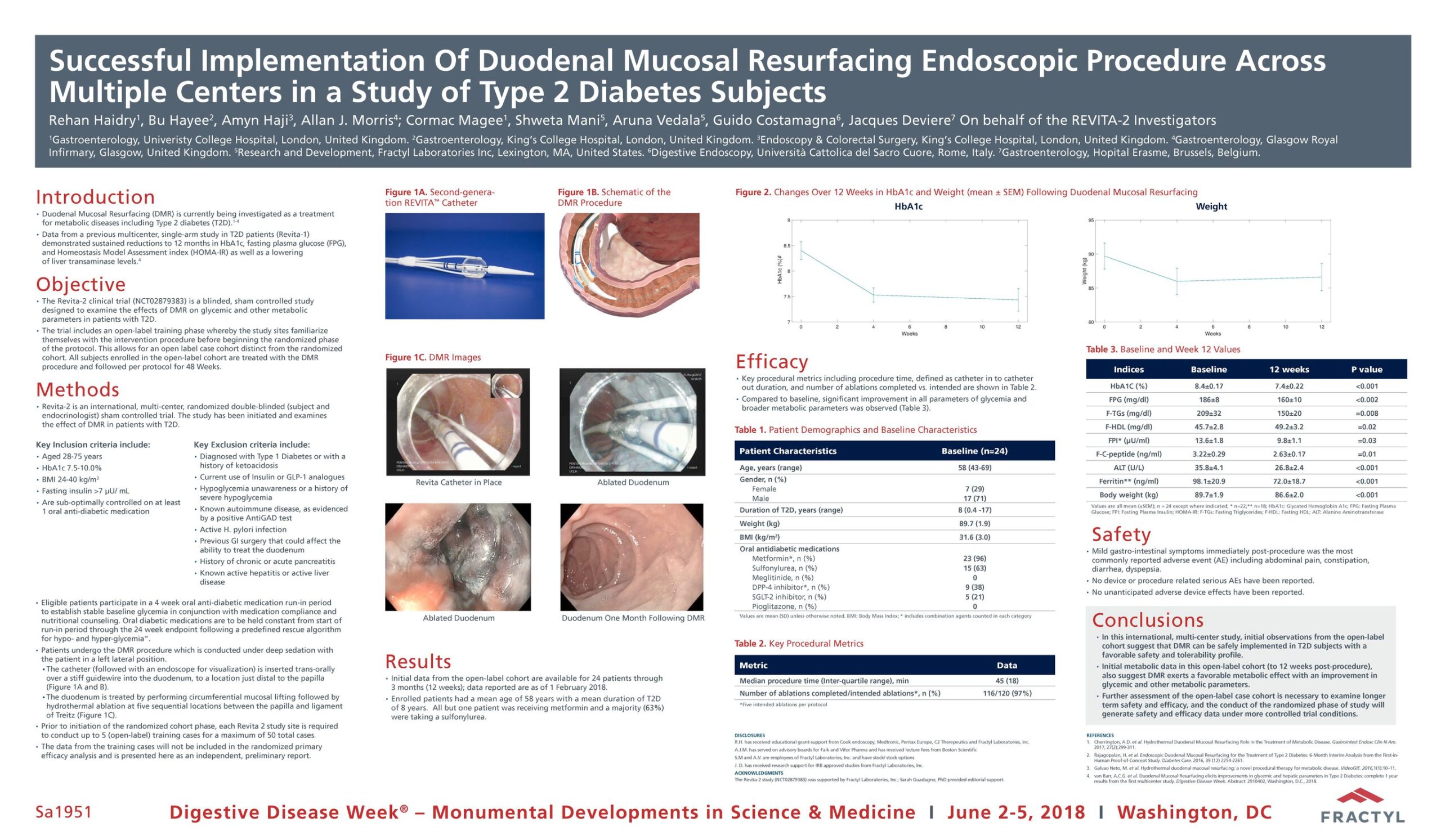
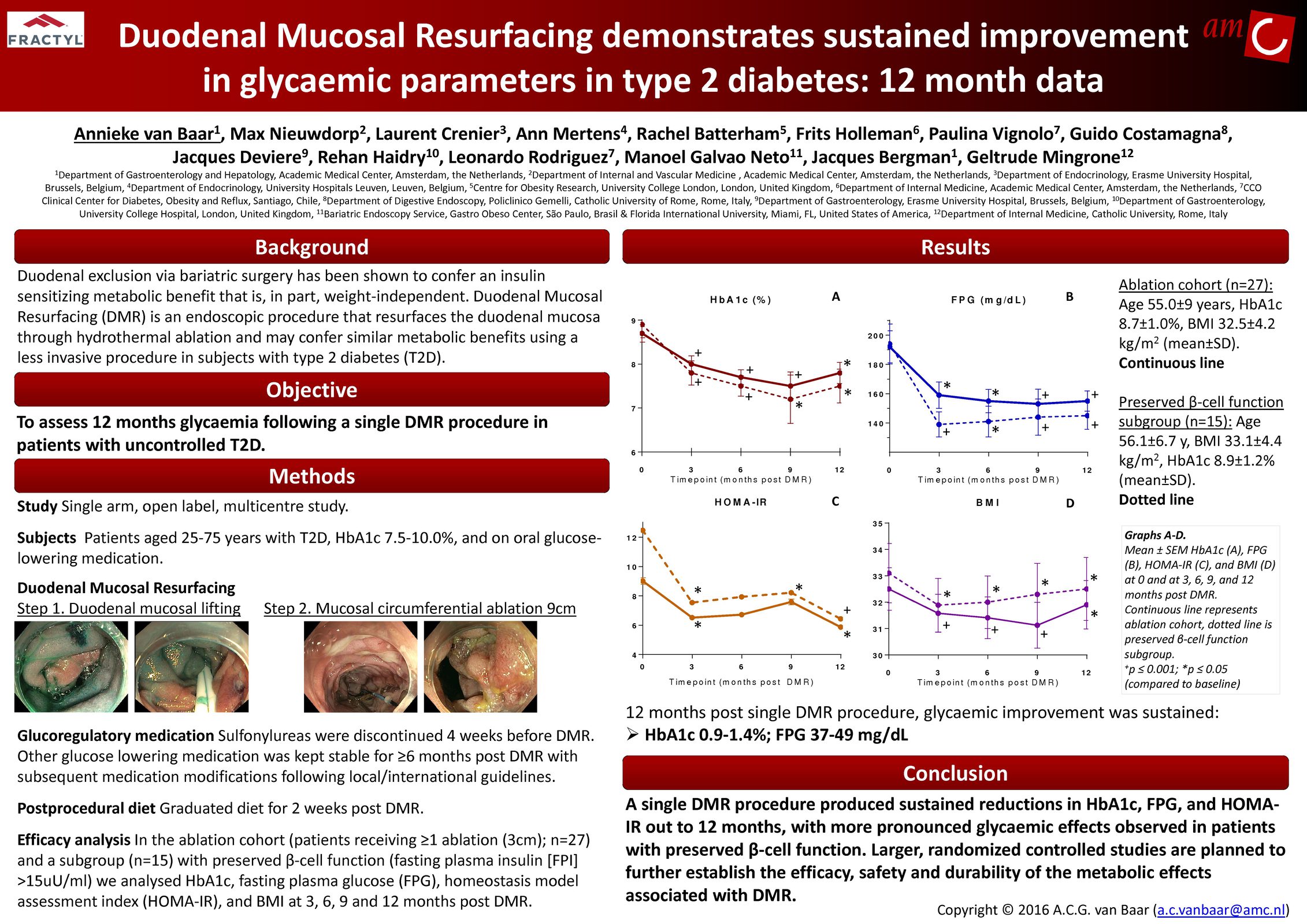
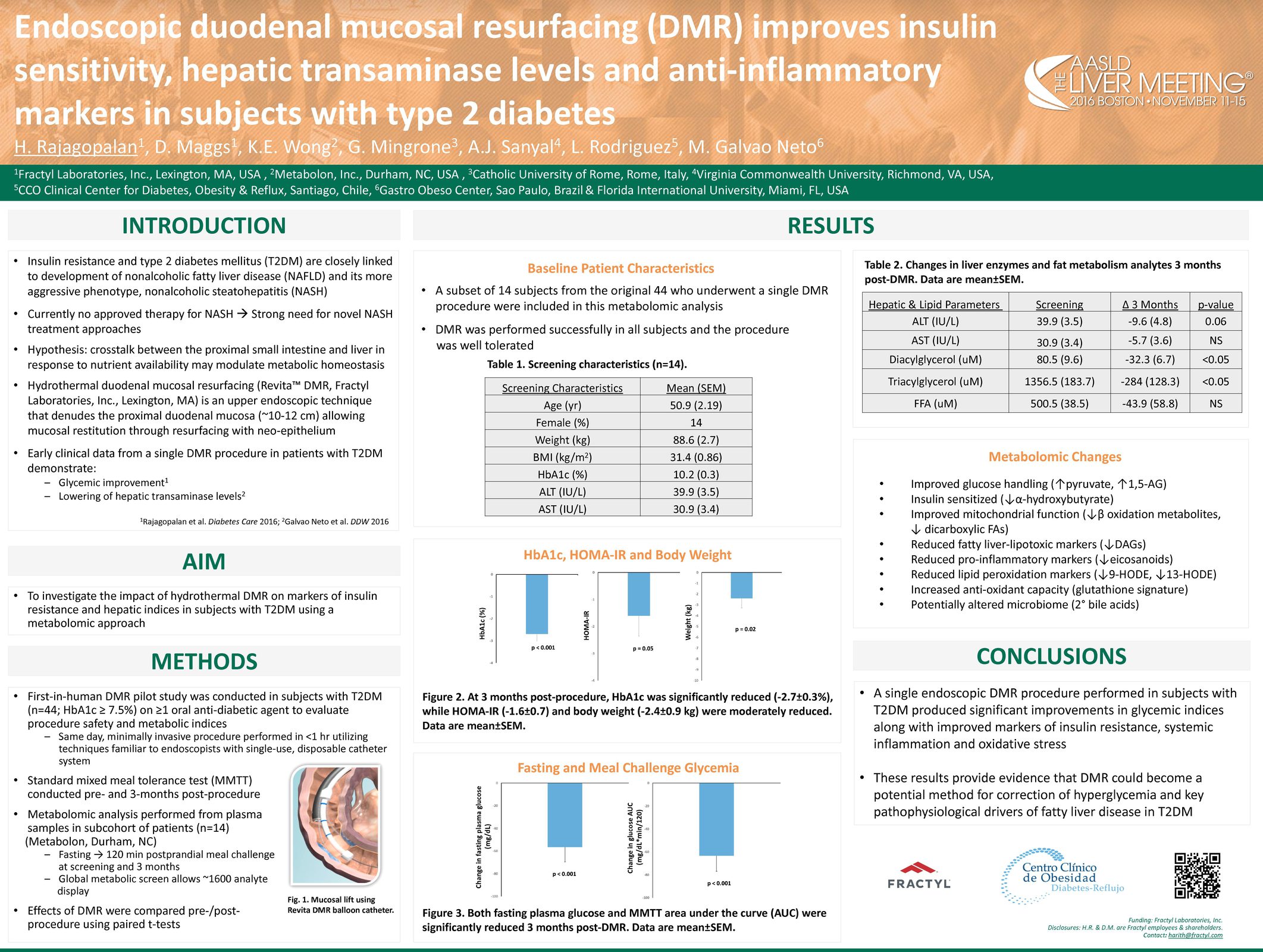
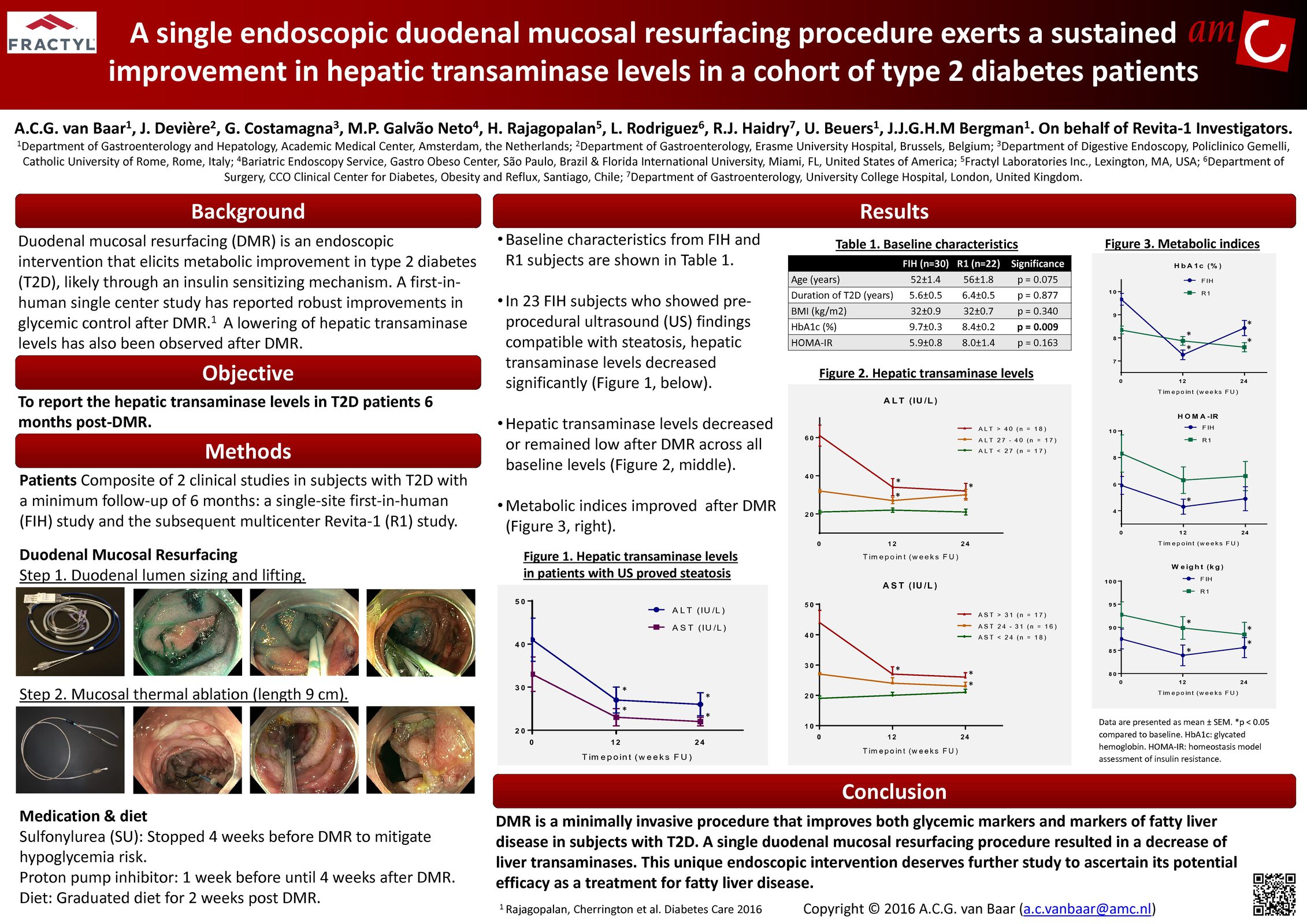
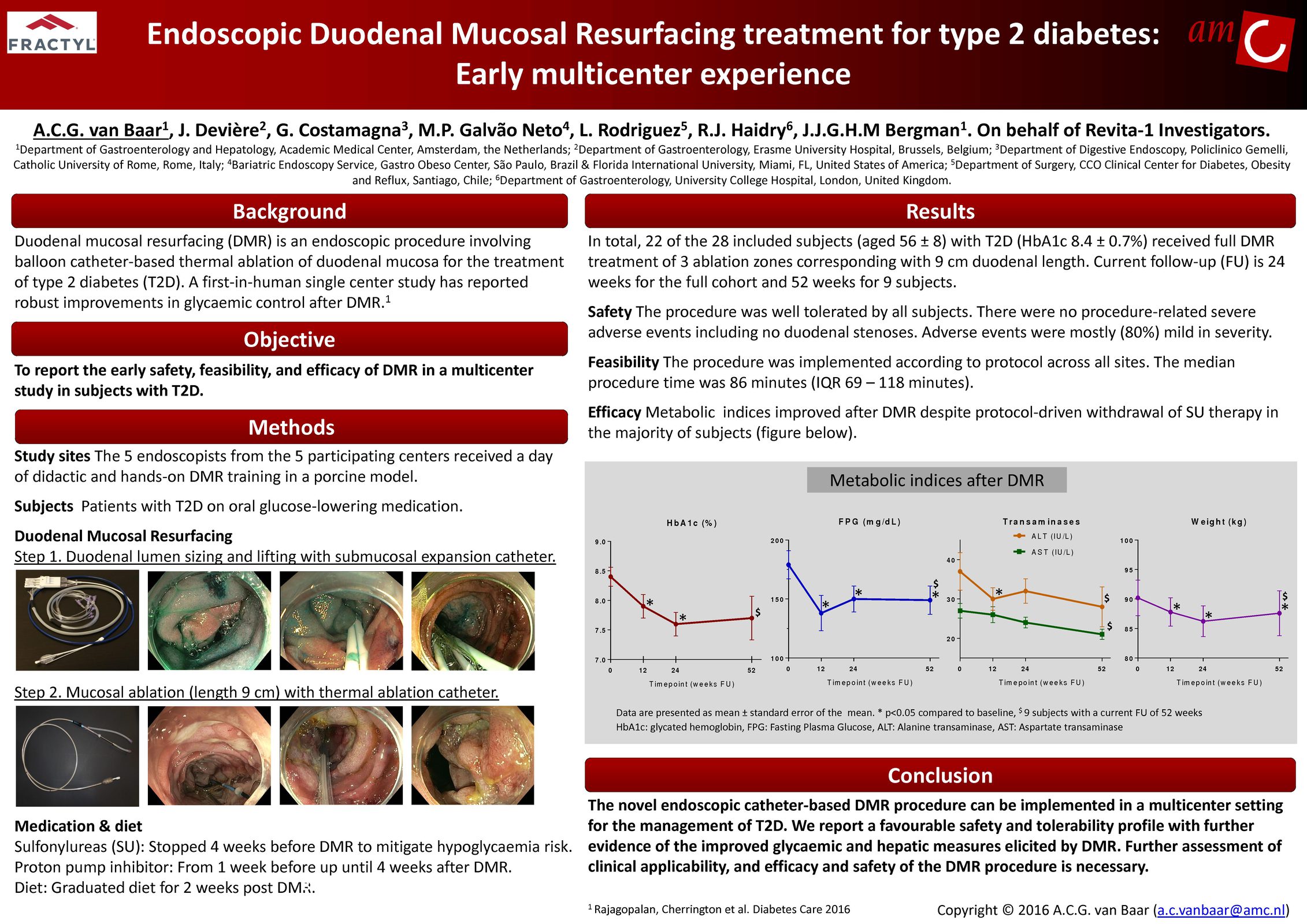
Advancing research on the role of the gut in metabolic disease
Led by world-renowned experts in metabolic disease, the EraseT2D task force shares Fractyl Health’s mission to build evidence, understand, and ultimately eradicate metabolic disease.
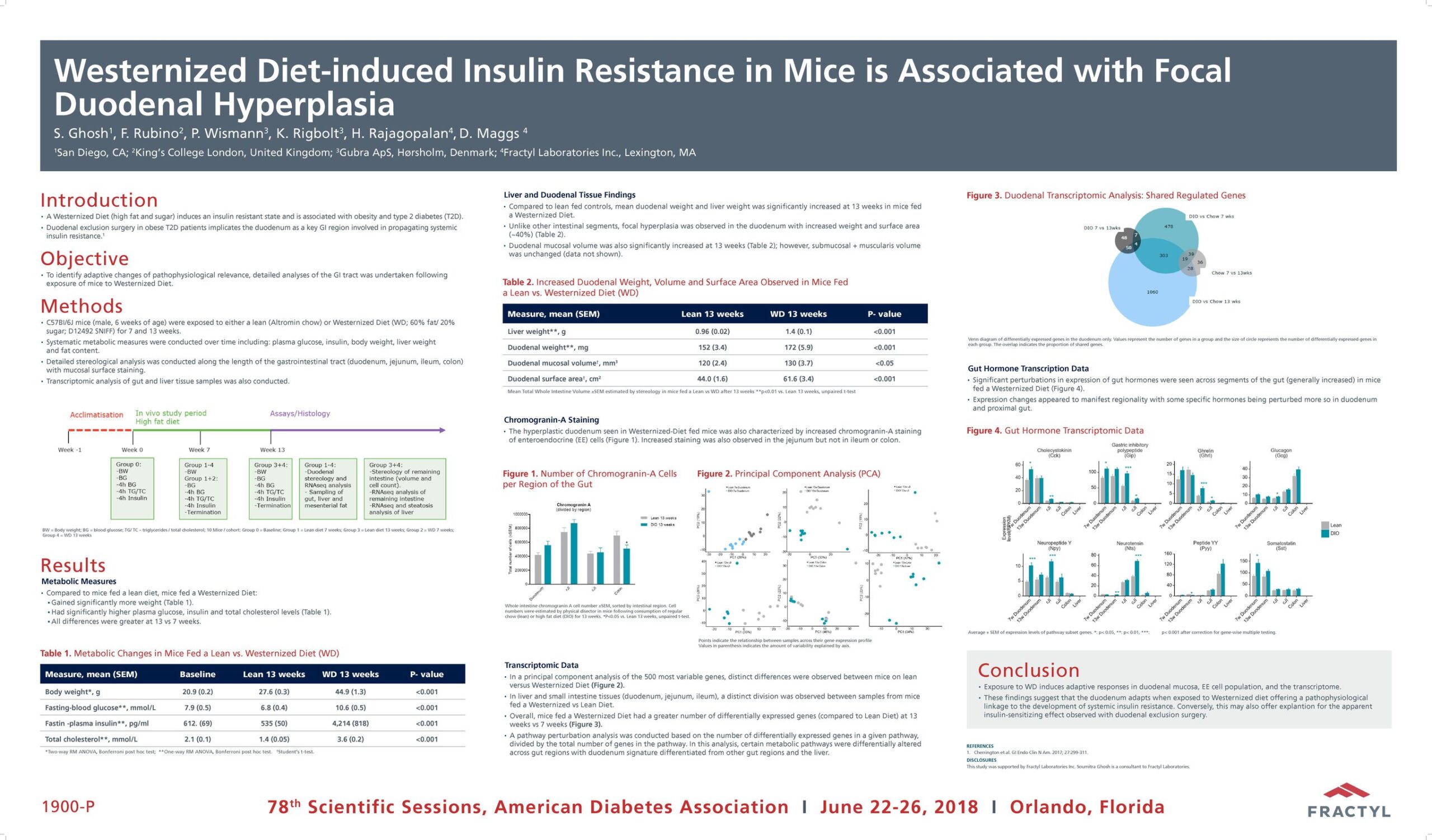
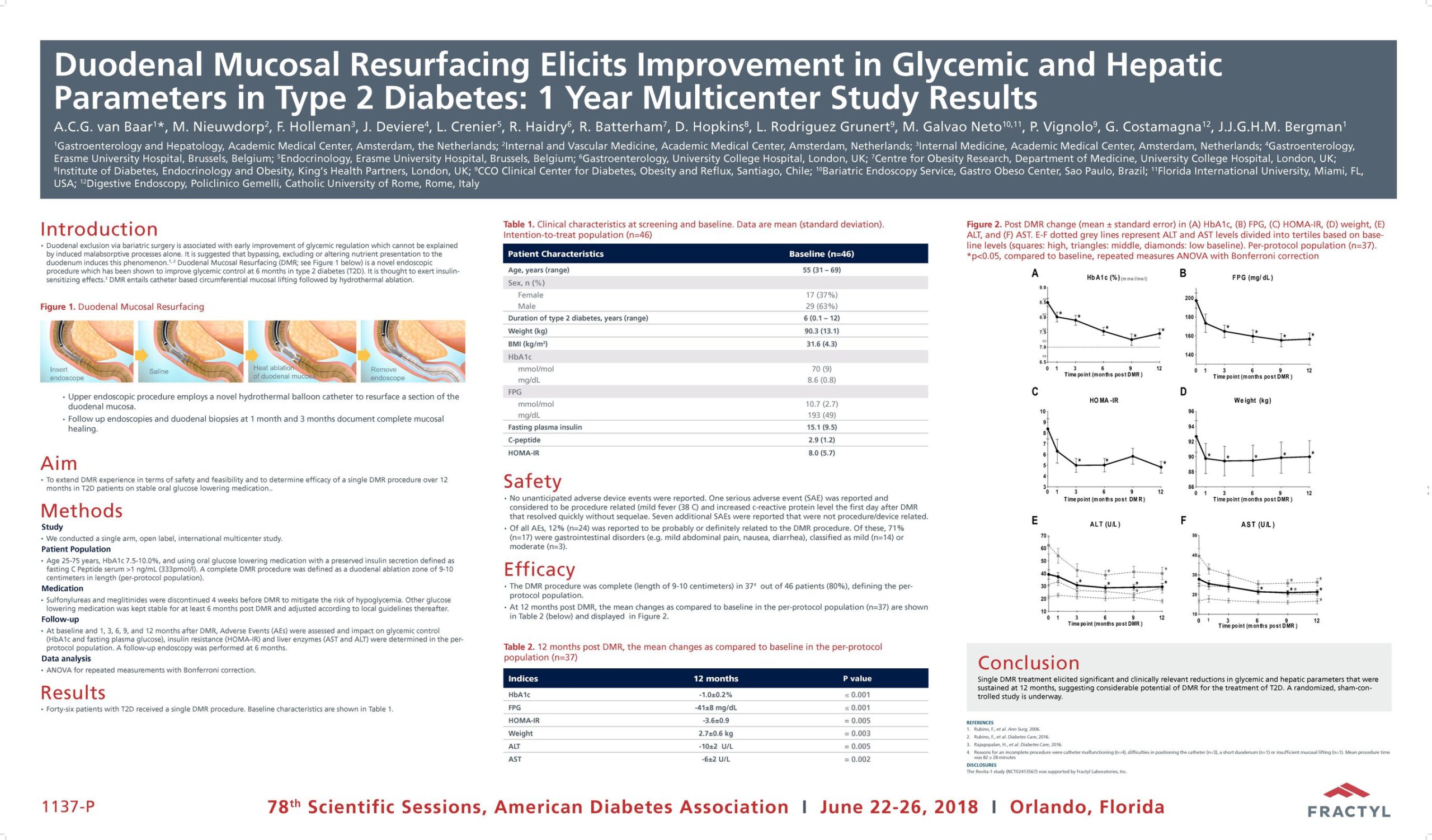
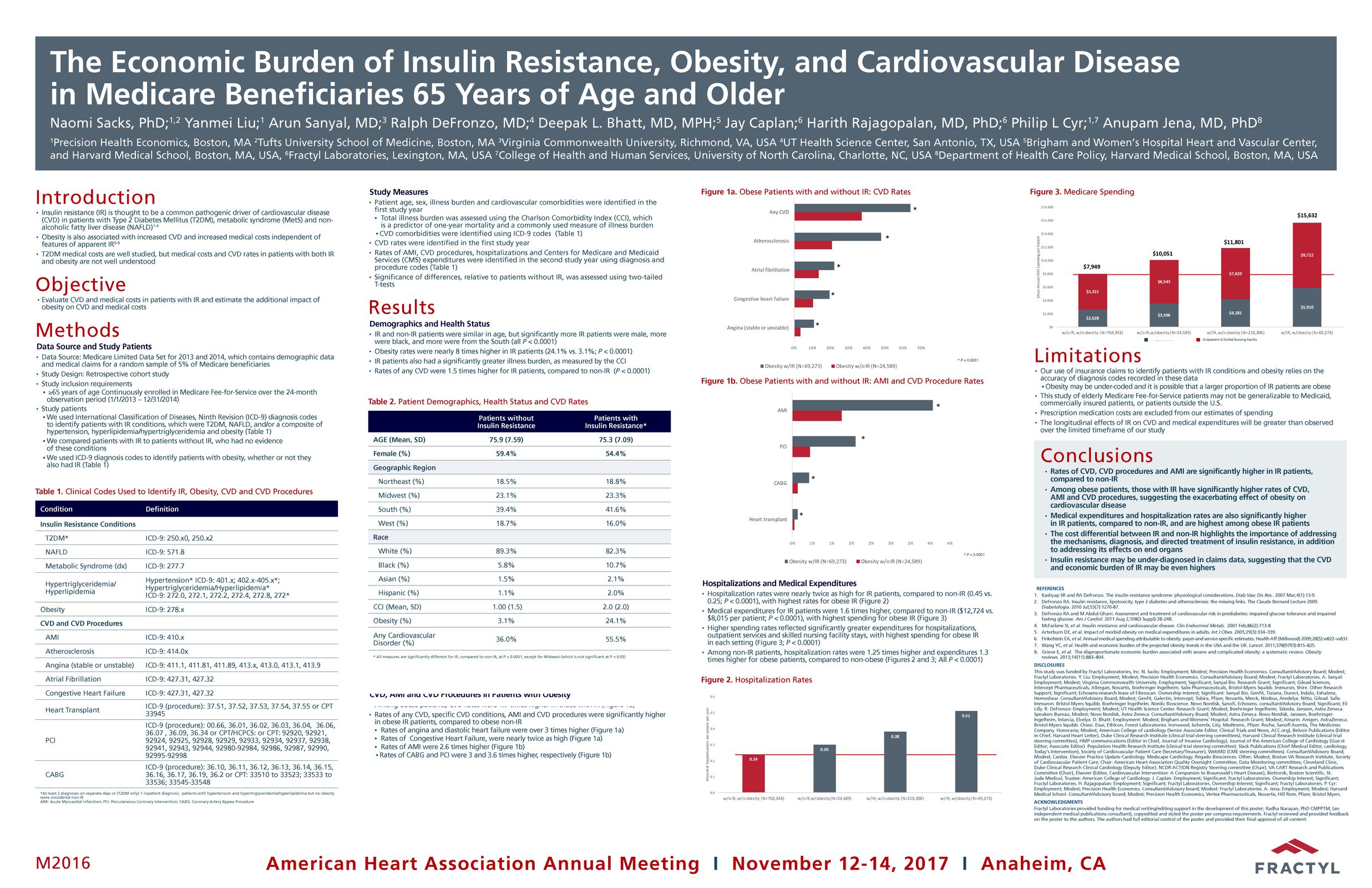
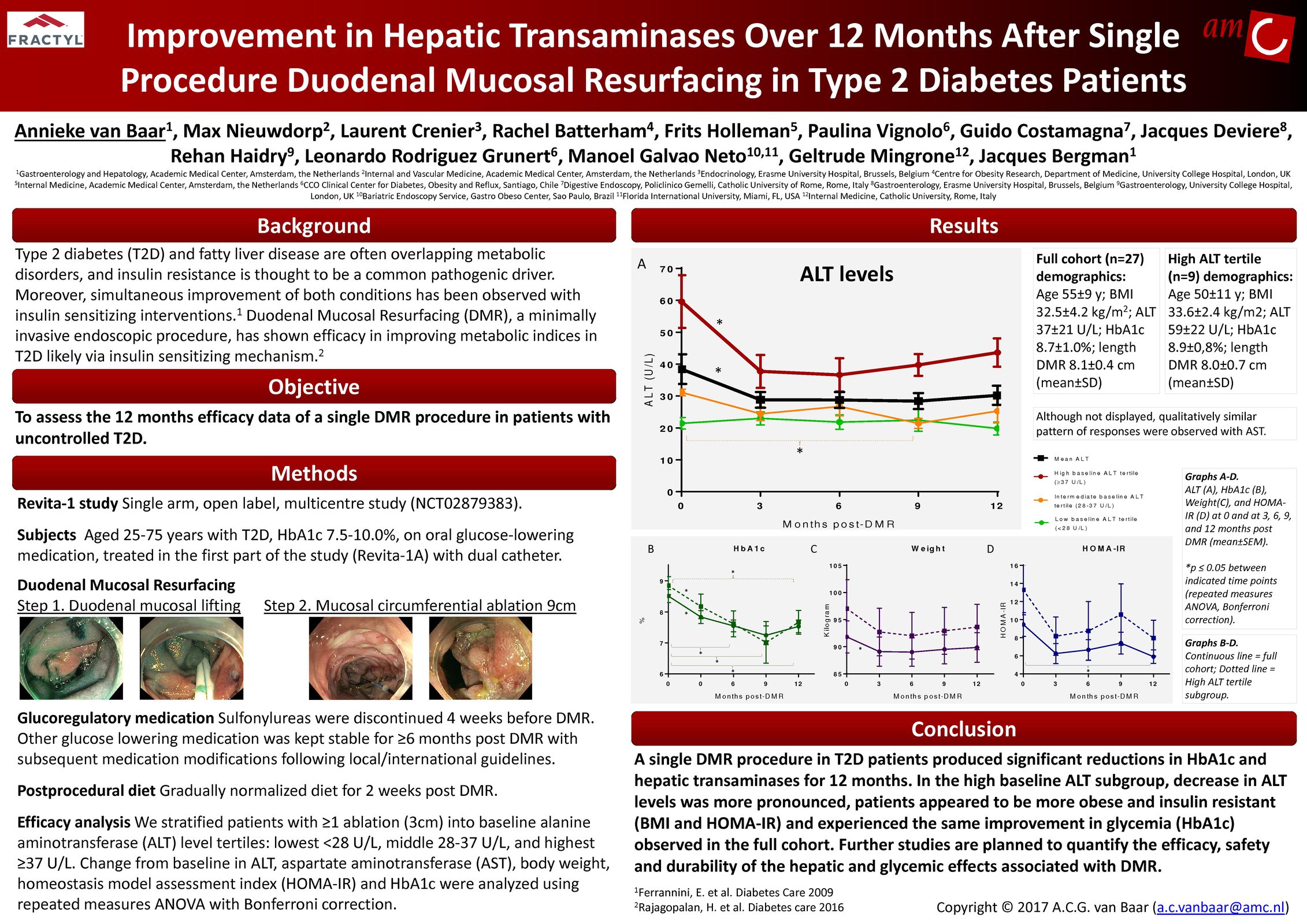
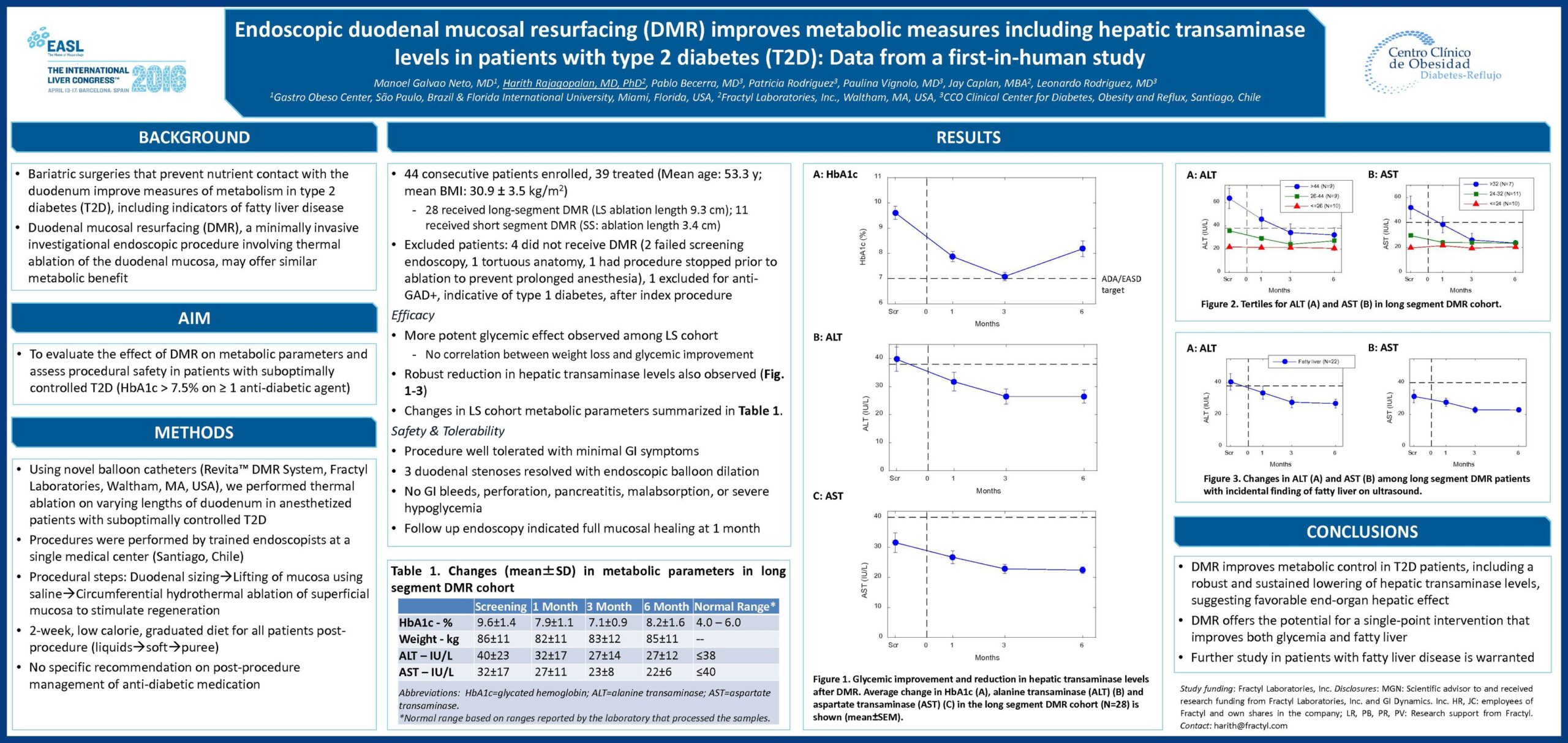
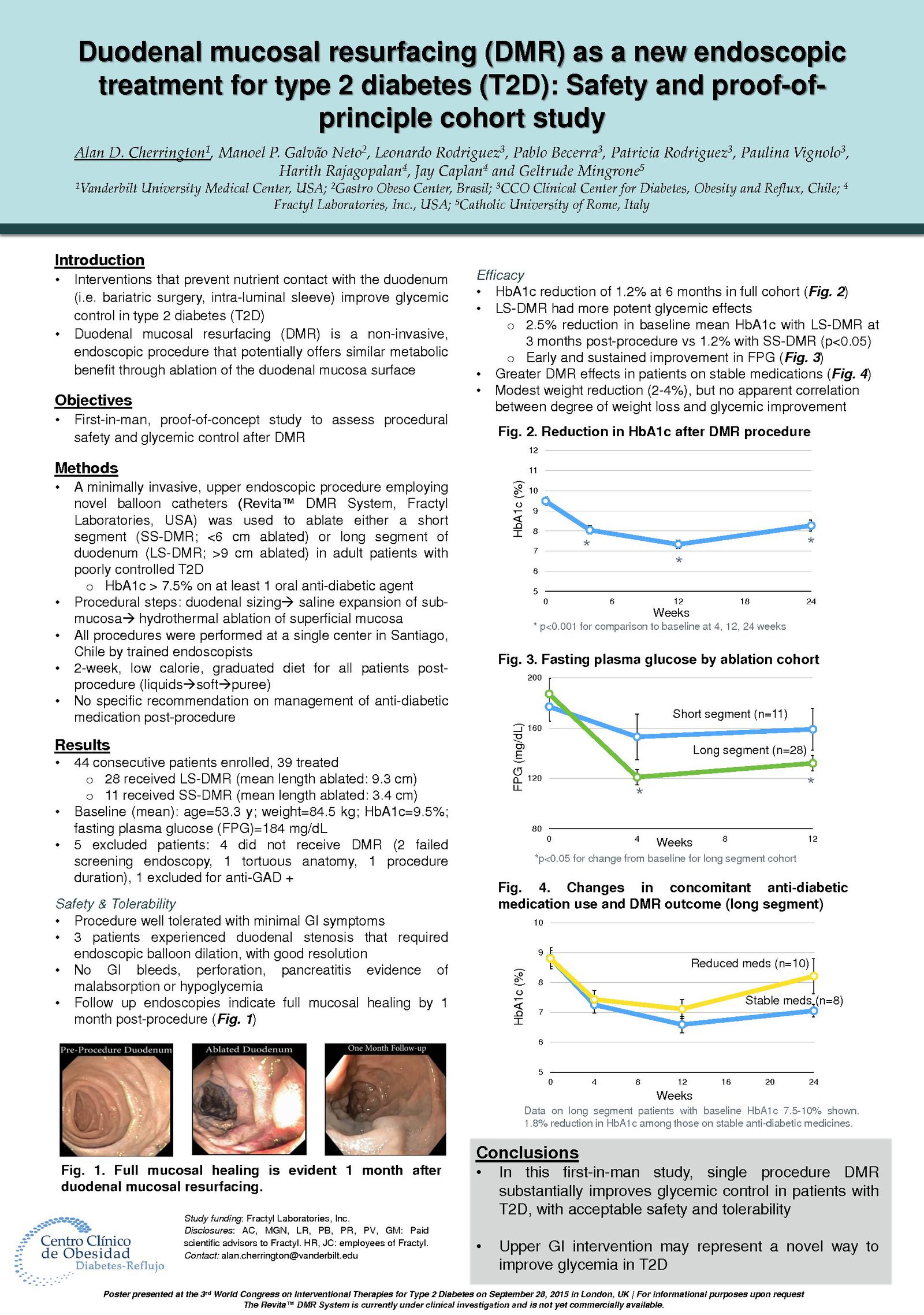
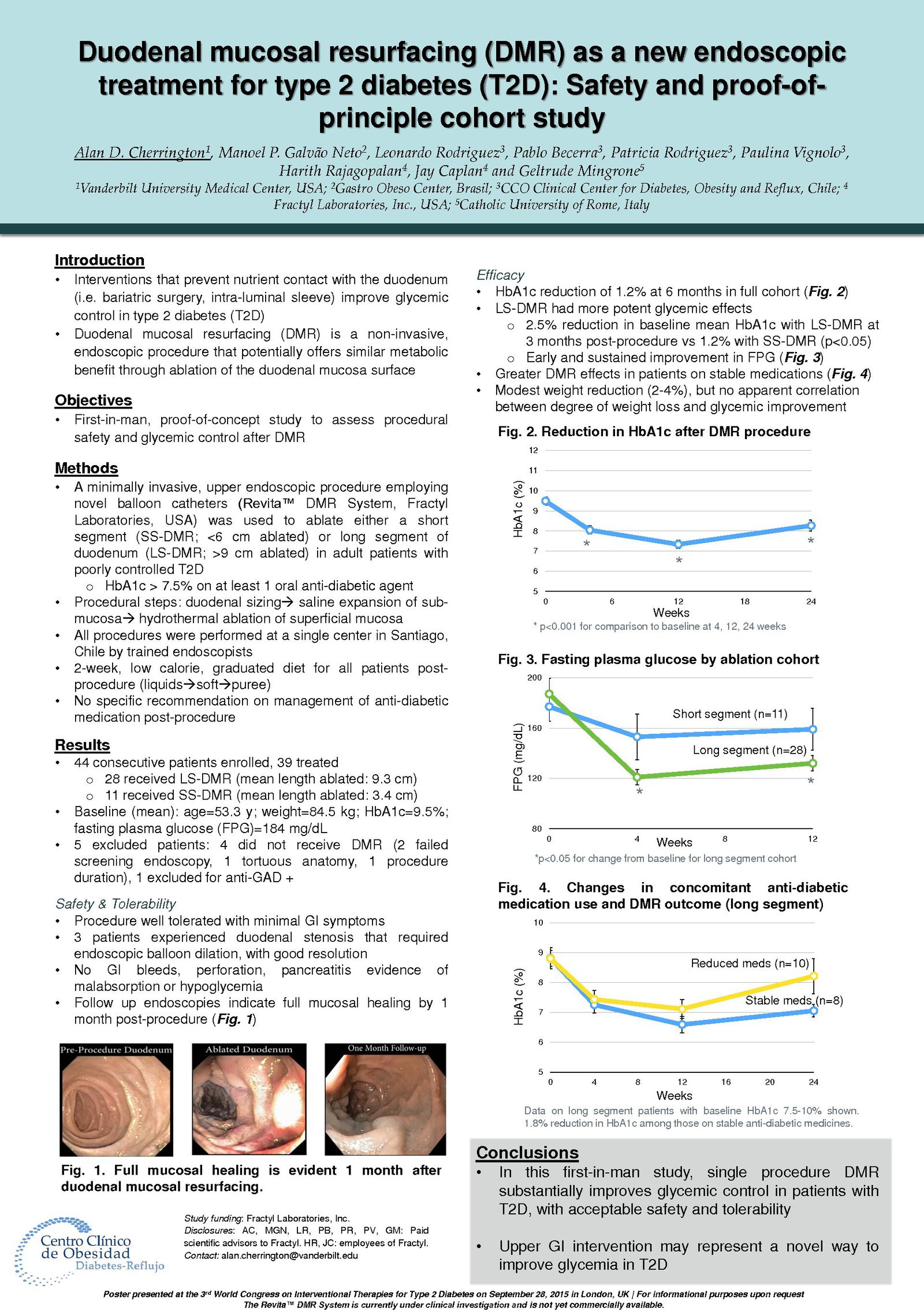
The Erase T2D task force builds on decades of discovery that have clearly identify the critical role of the gut as a regulator of metabolic disease. The signaling mechanisms between the gut and the rest of the body are numerous and not yet fully understood. The task force is charged with advancing research on the role of the gut in metabolic disease to catalyze future discoveries that may inform how diabetes can be better understood and ultimately erased.
This initiative, funded by Fractyl Health, is meant to spur the kind of innovative thinking and fundamental breakthroughs that will point the way toward a world without type 2 diabetes.
The ERASE T2D Task Force is comprised of the following members: