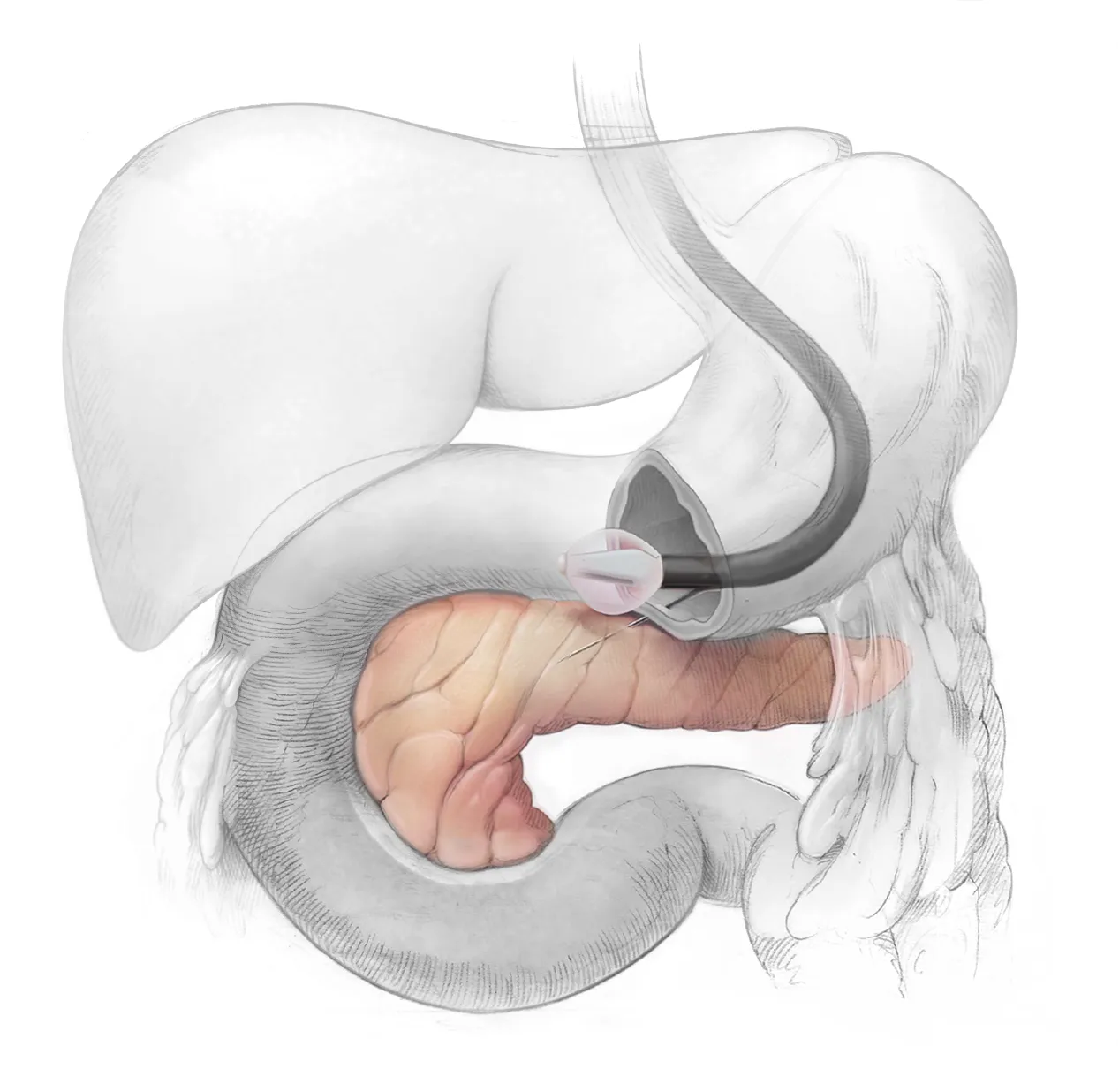
Rejuva is a novel, locally administered AAV gene therapy platform in preclinical development aimed at enhancing islet function. Designed as a one-time treatment, it represents a major advance in incretin-based therapies by reprogramming metabolic hormone response in pancreatic islet cells to drive durable weight loss and long-term remission of type 2 diabetes.
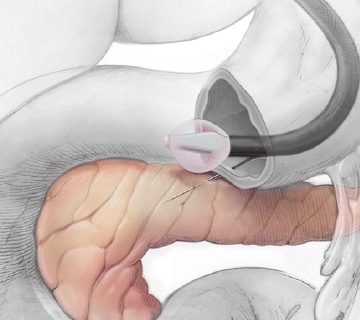
Proprietary delivery system to enable local delivery of advanced therapies directly to pancreas.
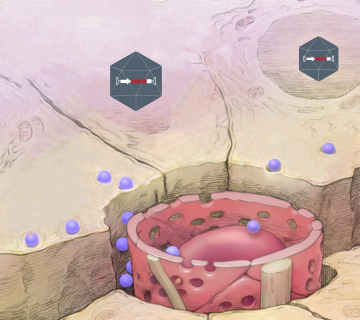
Low dose viral vectors with high transduction efficiency and limited systemic biodistribution.
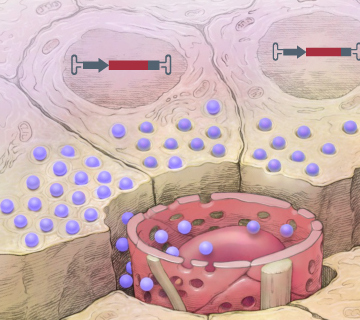
Durable alteration of metabolic hormone response in the islets with tissue-restricted transgenes.
Rejuva® is in early development and not approved by any regulatory body for investigational or commercial use.