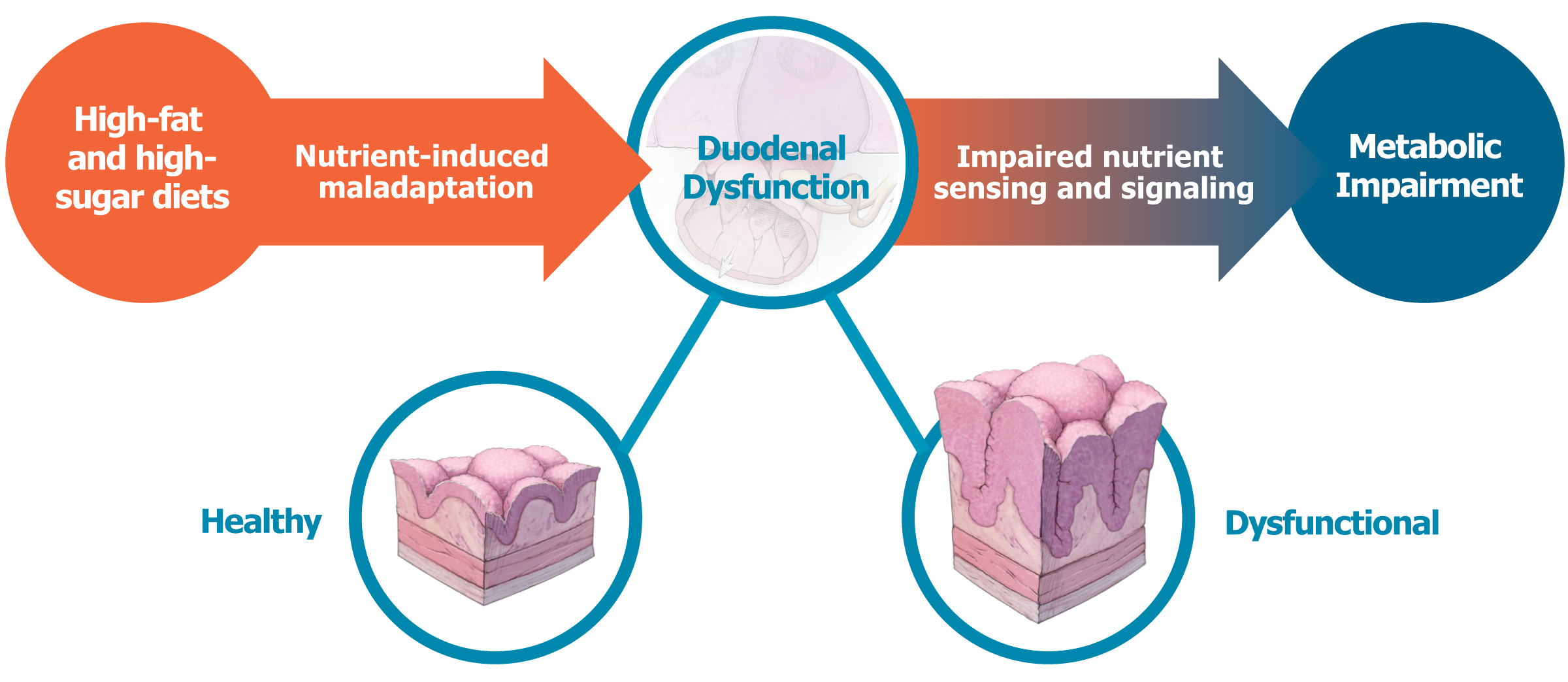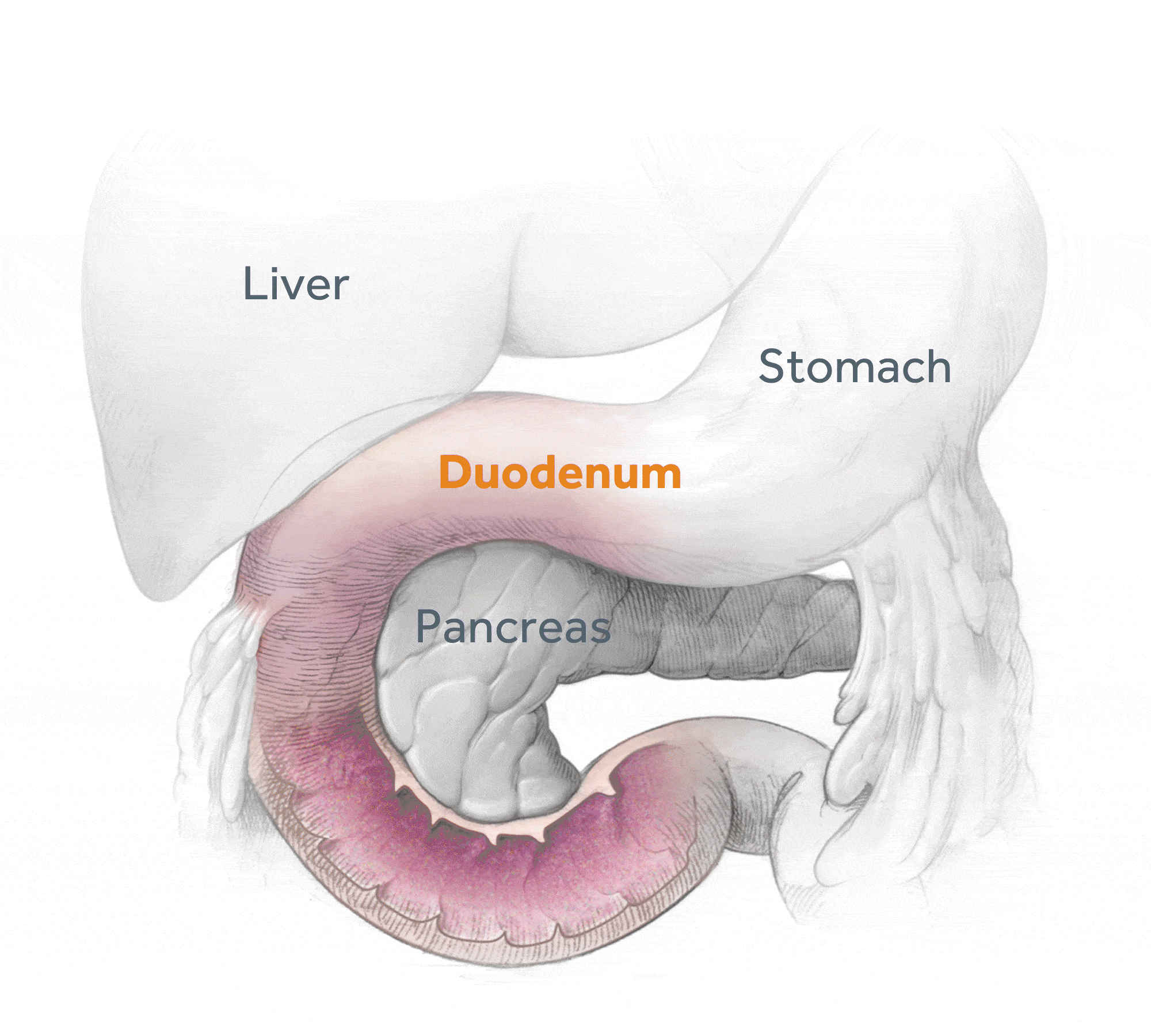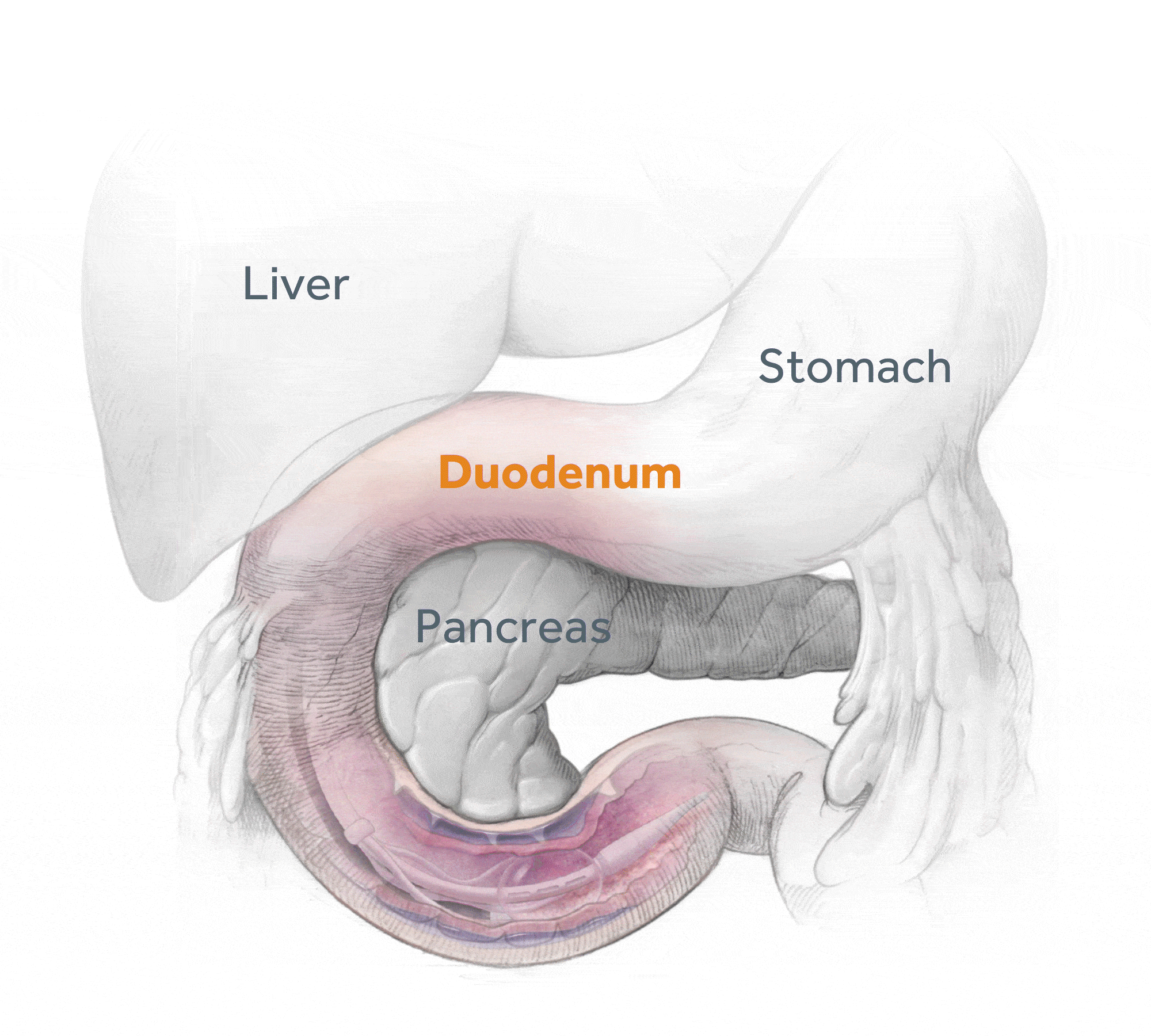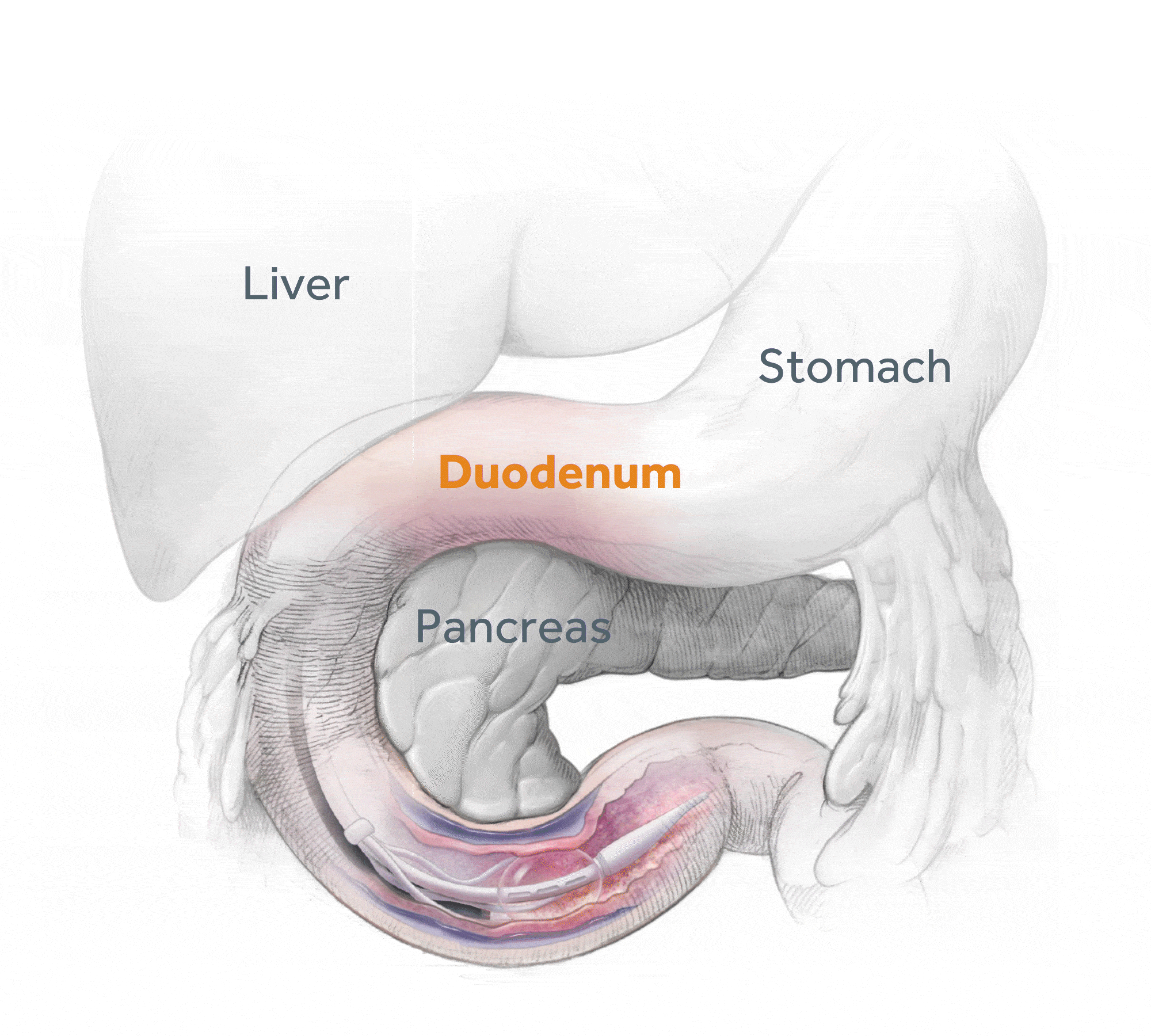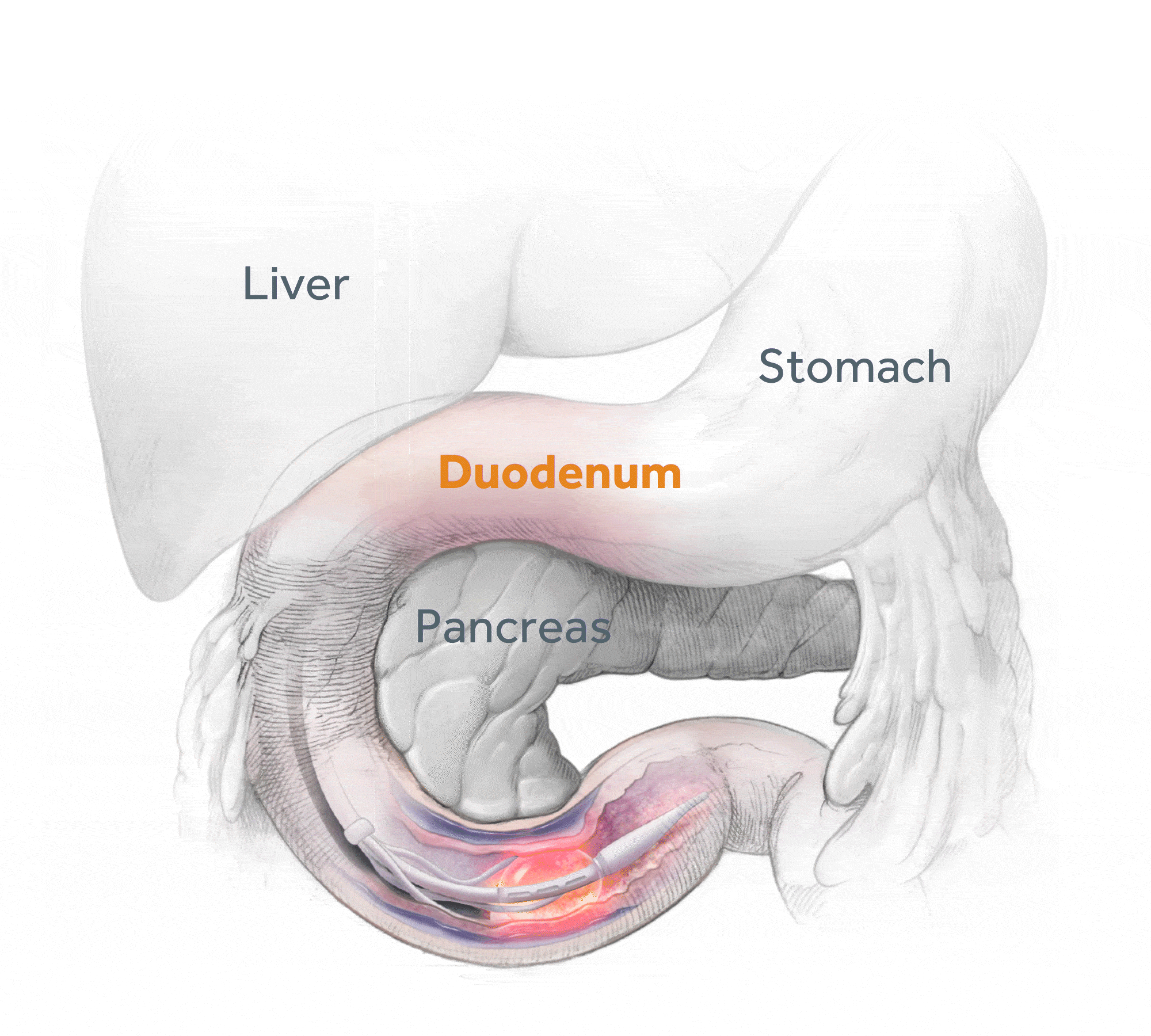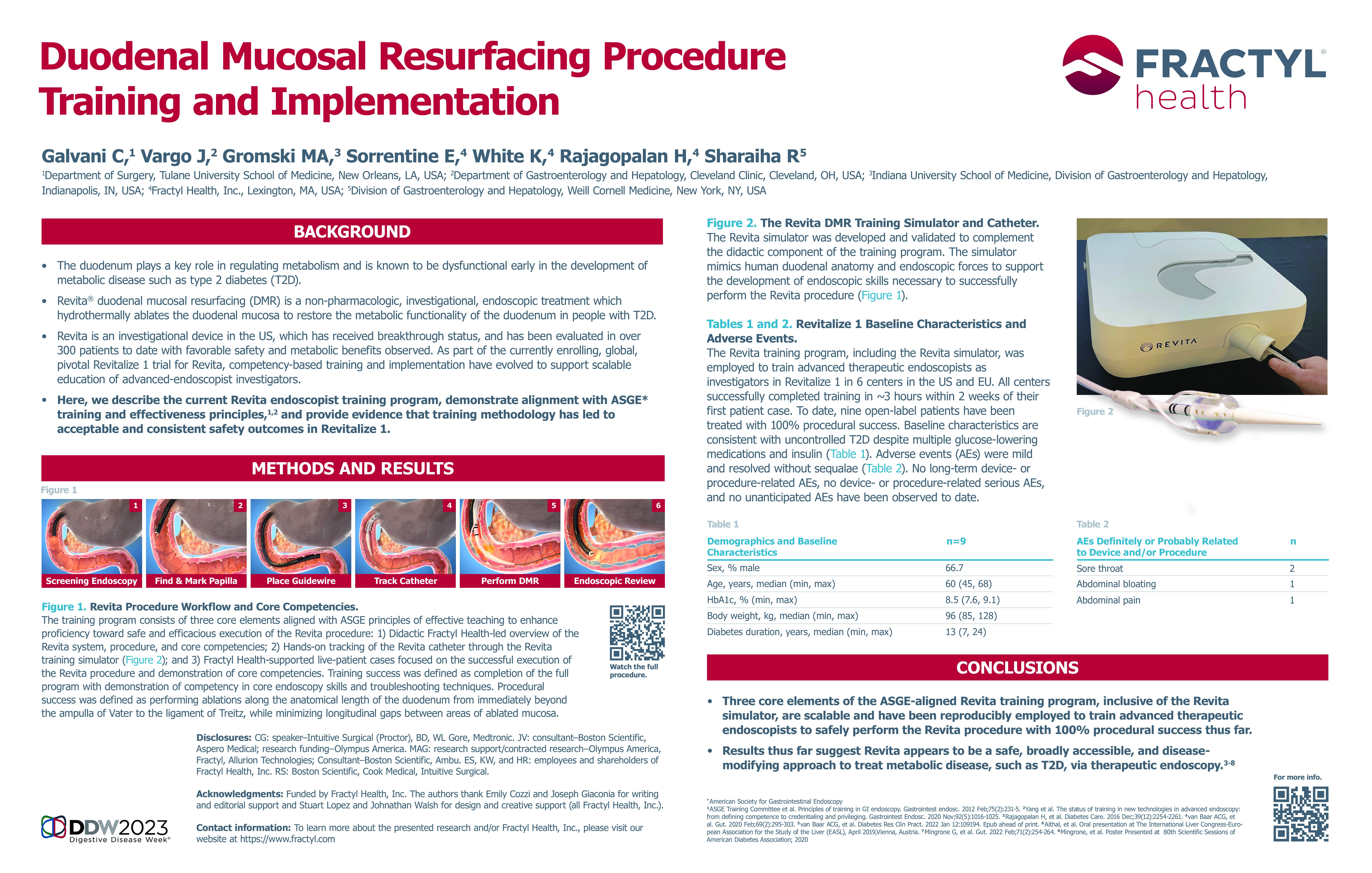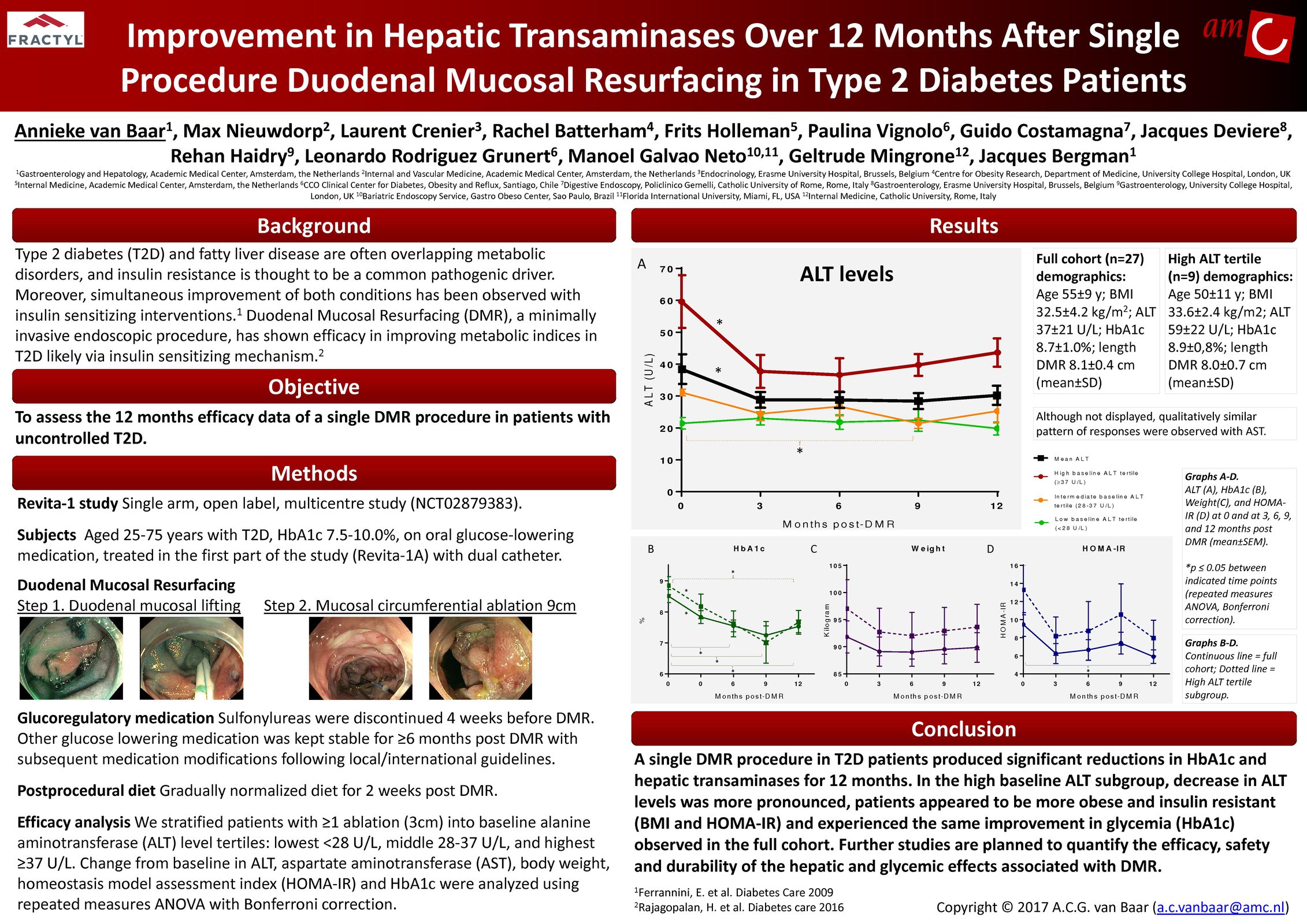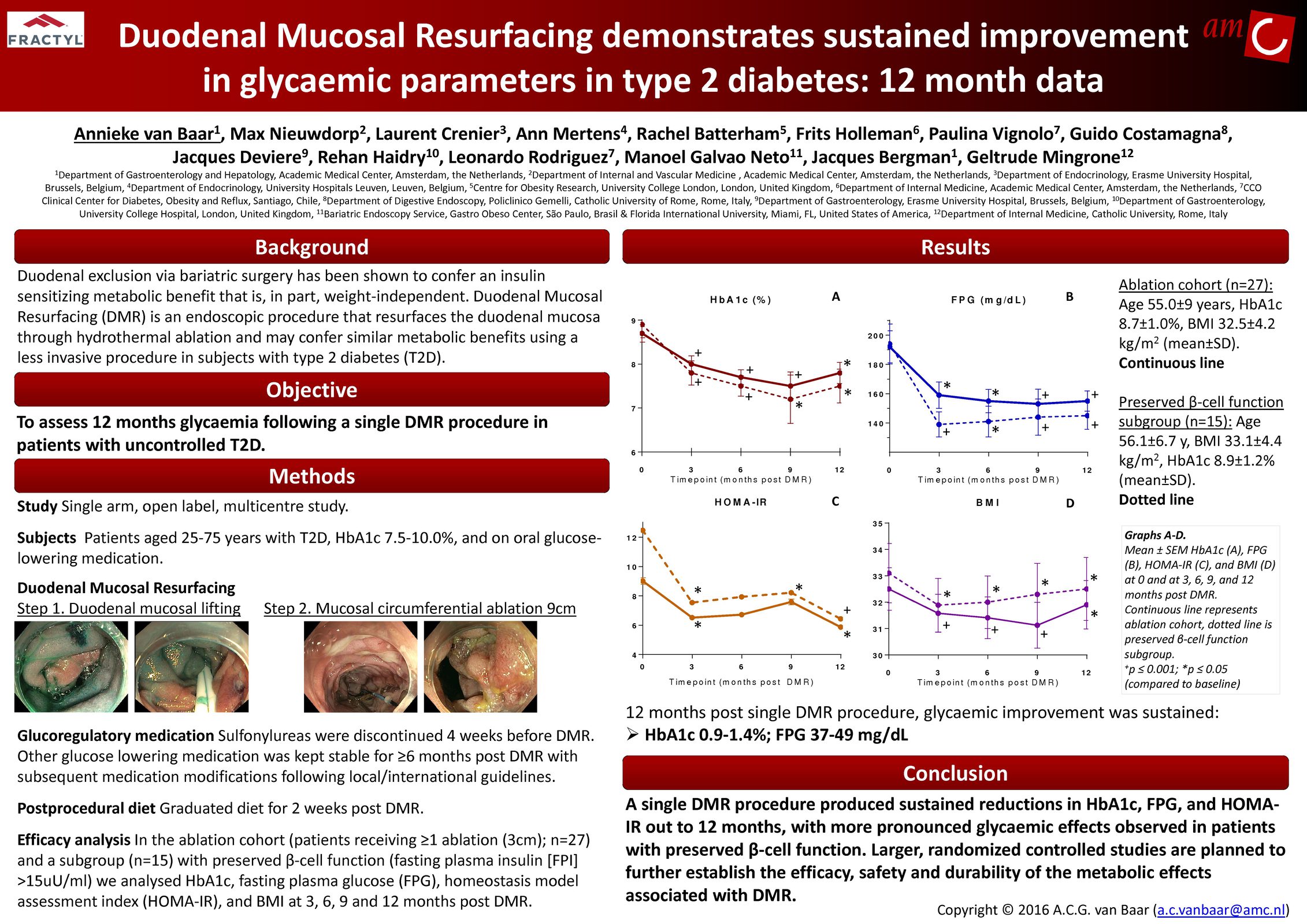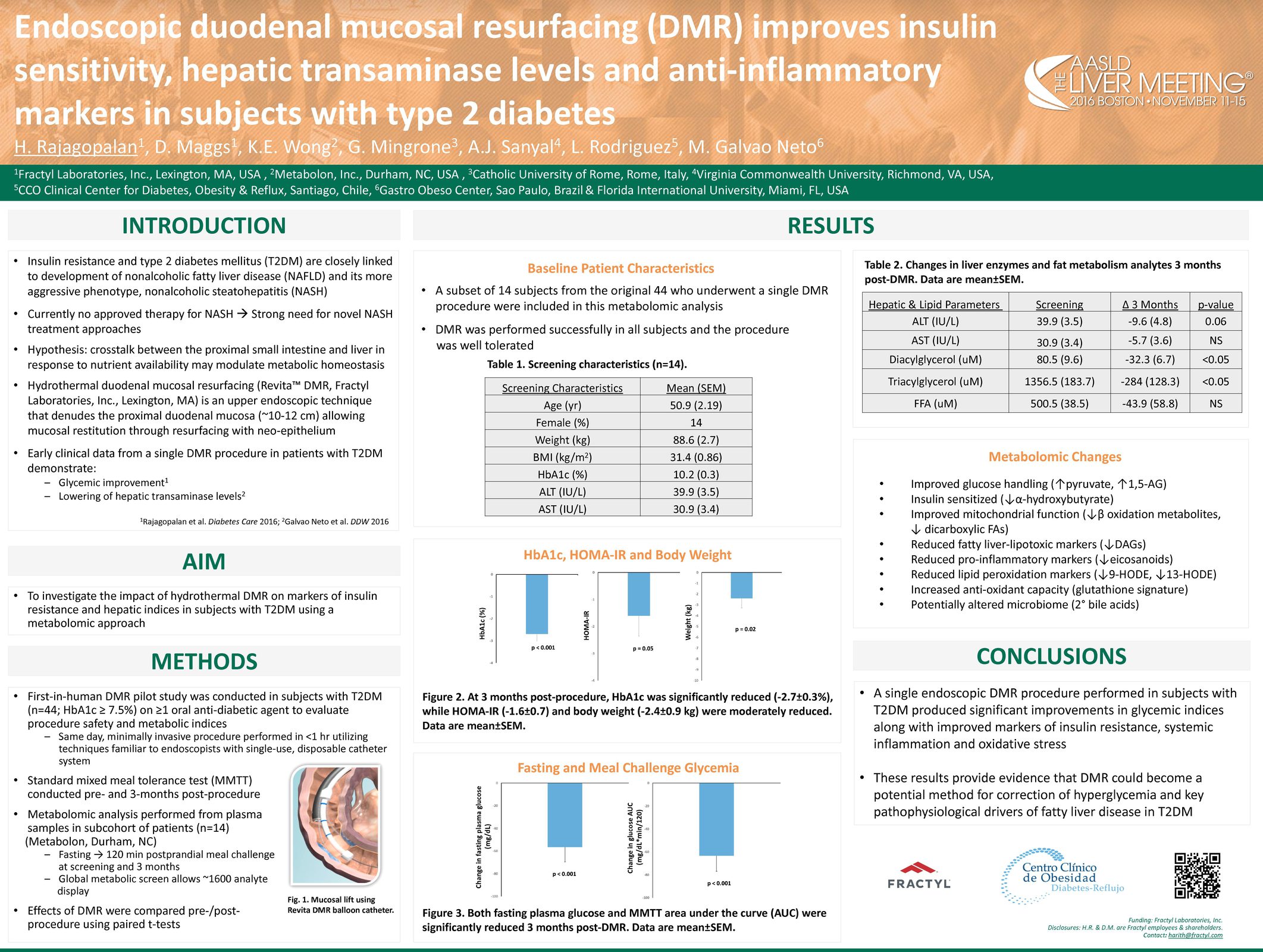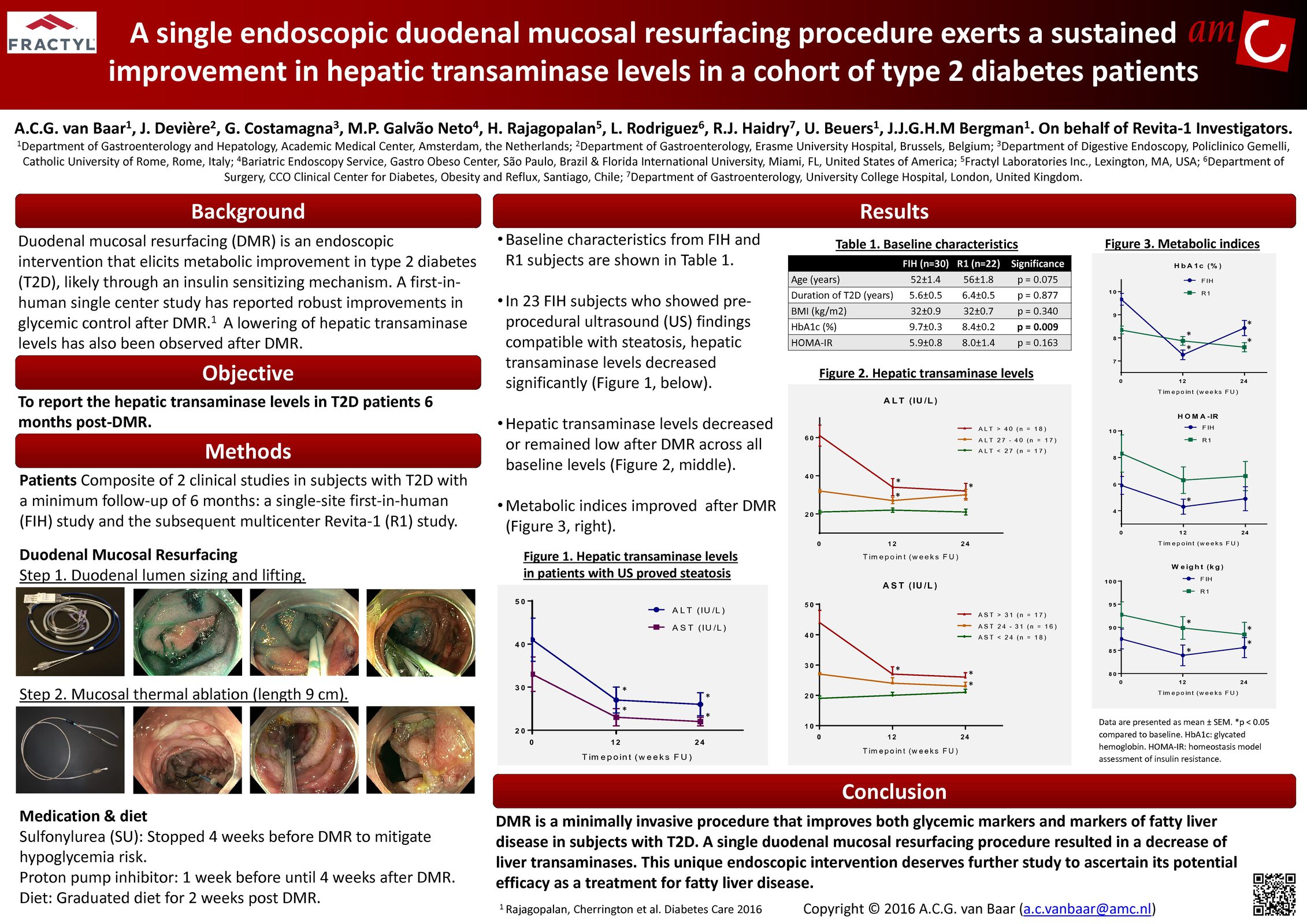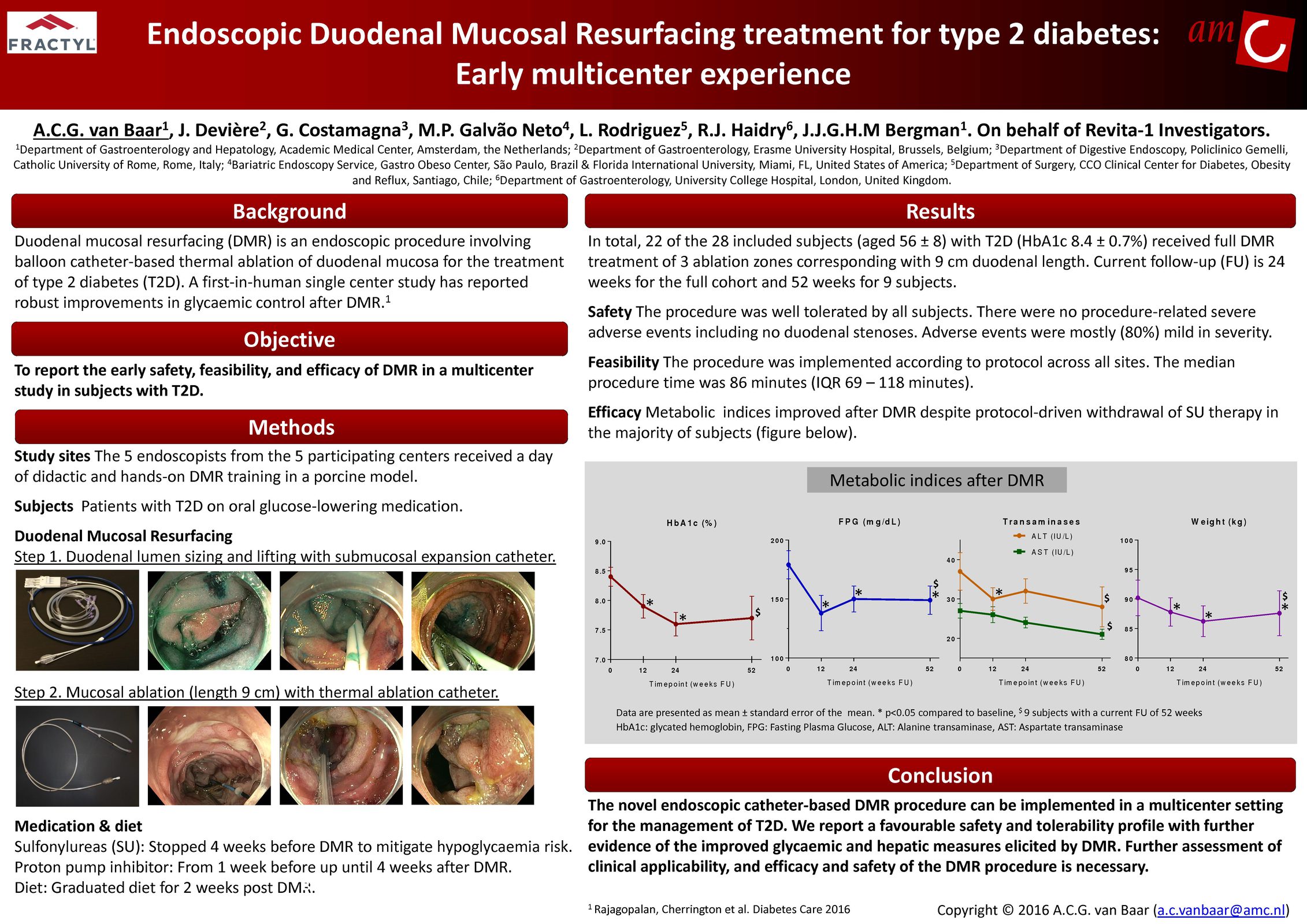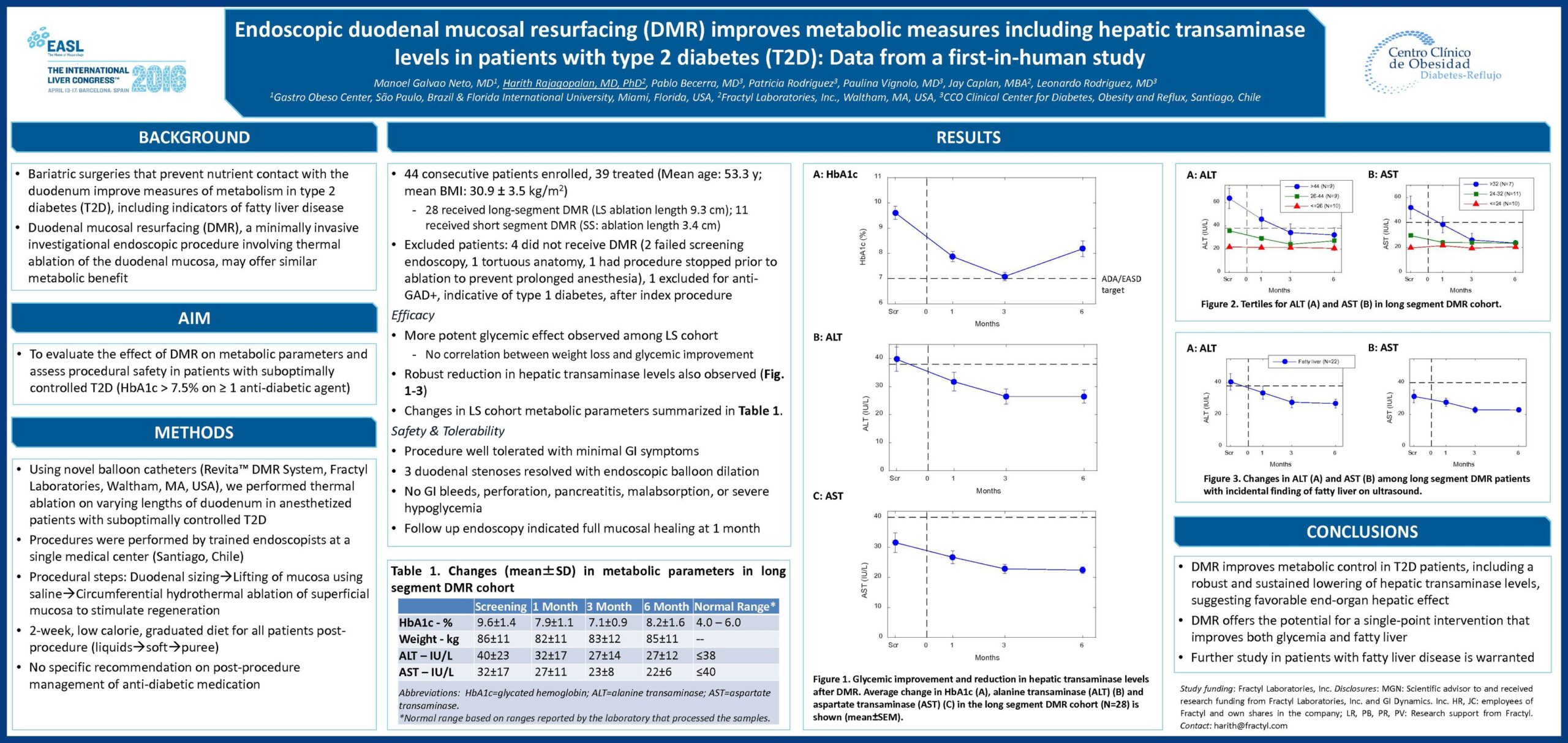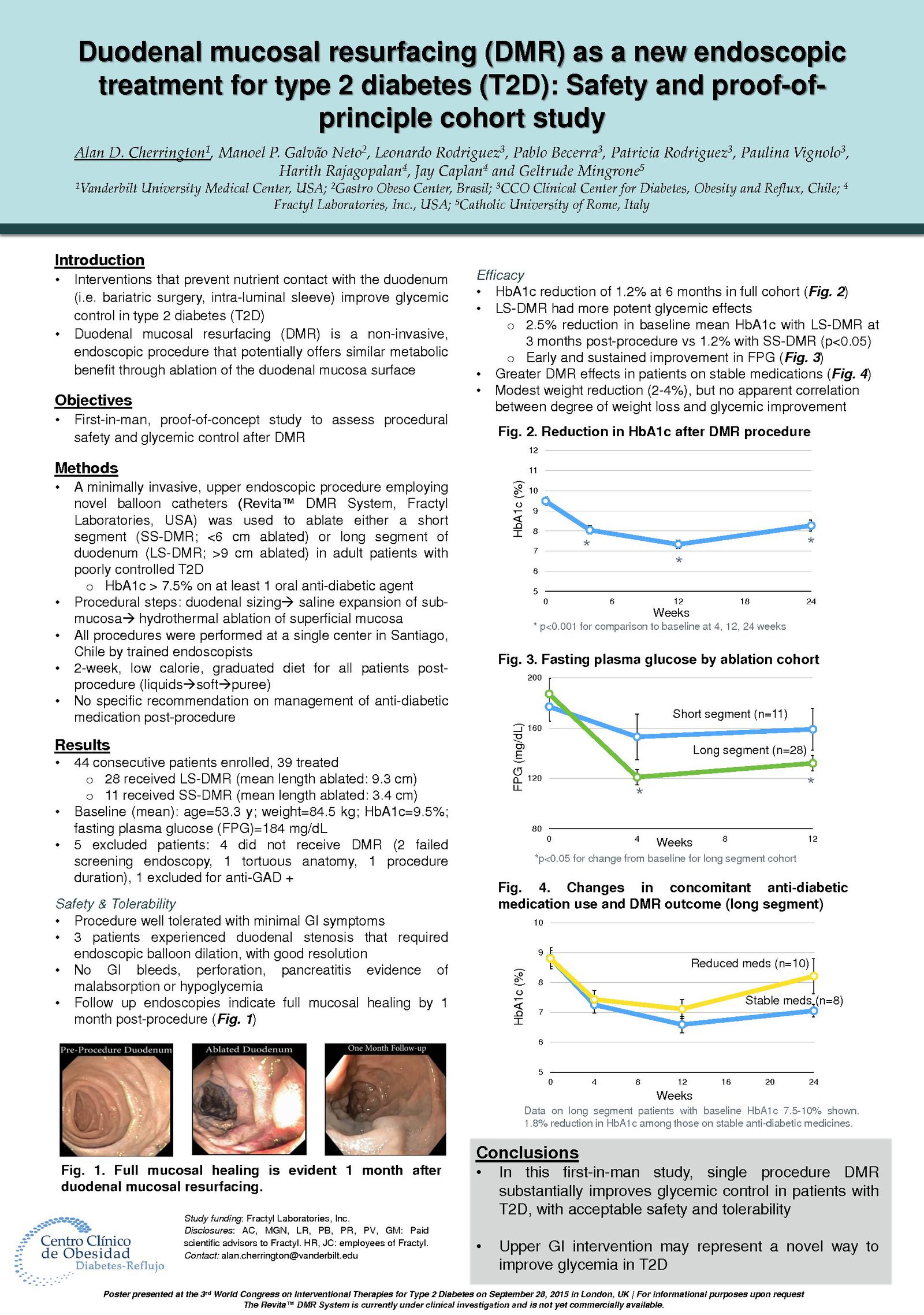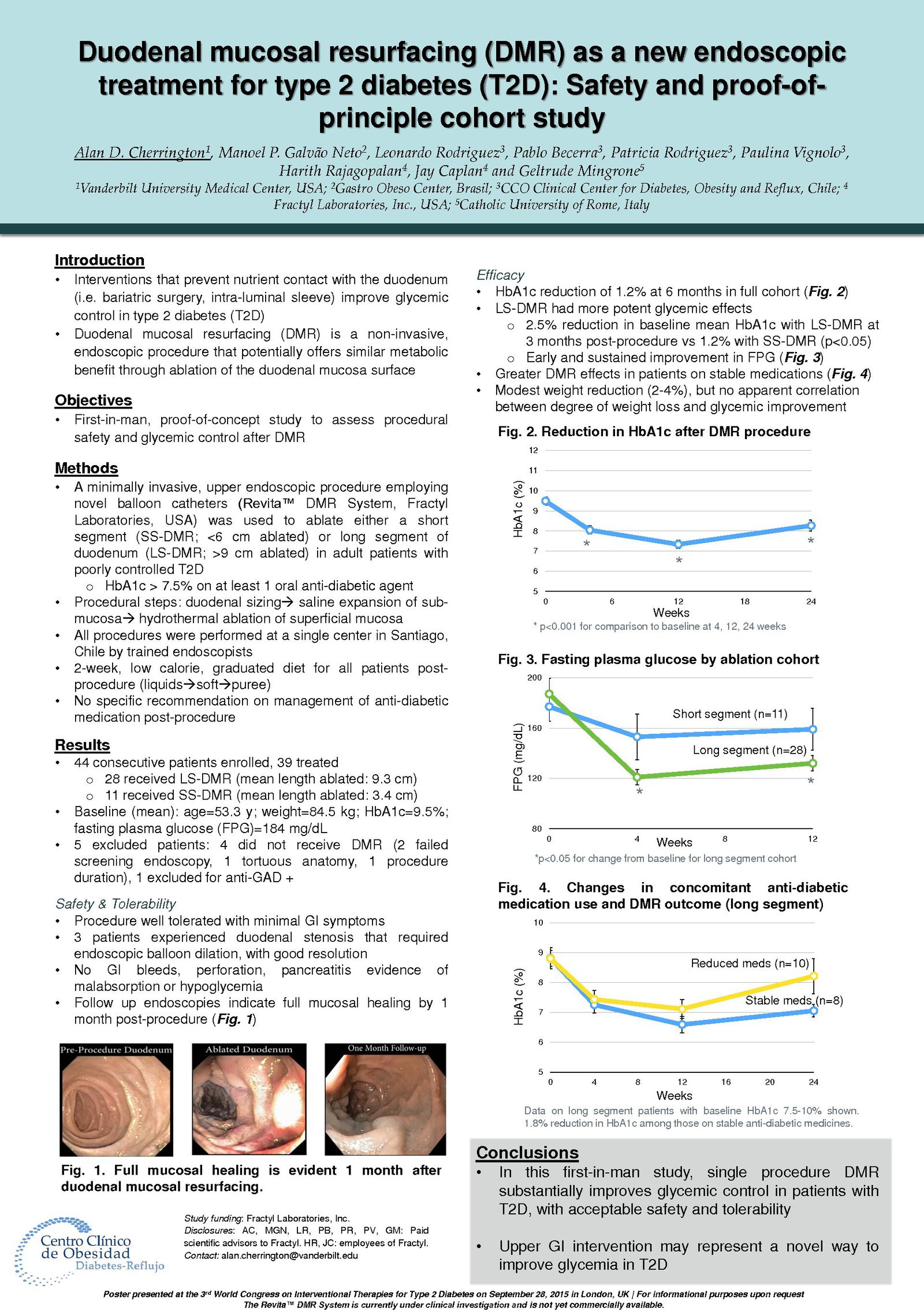
Glycemic Improvement, Insulin Reductions, and Improved Body Weight 48 Weeks after Revita Duodenal Mucosal Resurfacing in T2D patients with Previously Inadequately Controlled Glucose Despite Multiple Glucose-Lowering Agents Including Insulin
Fonseca, et al. presented poster at American Diabetes Association 2023, San Diego, CA, June 2023
View Poster